
��8�֣�̼�ǻ�������������Ԫ�أ��䵥�ʼ������������������������Ҫ��Դ���ʡ�
��1��ʹCl2��H2O(g)ͨ�����ȵ�̿�㣬����HCl��CO2������1molCl2���뷴Ӧʱ�ͷų�145kJ������д���÷�Ӧ���Ȼ�ѧ����ʽ�� ��
��2����ѧ������̫���ֽܷ�ˮ���ɵ������ڴ����������������̼��Ӧ���ɼ״�����֪H2(g)��CO(g)��CH3OH(l)��ȼ���Ȧ�H�ֱ�Ϊ��285.8kJ��mol-1����283.0kJ��mol-1�ͣ�726.5kJ��mol-1����
����̫���ֽܷ�10molˮ���ĵ�������_____________kJ��
�ڼ״�����ȫȼ������һ����̼��Һ̬ˮ���Ȼ�ѧ����ʽΪ_______ ____ ��
��3����֪��Fe2O3(s)��3C(ʯī)��2Fe(s)��3CO(g) ��H����489.0 kJ��mol-1
CO(g)��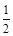 O2(g) ��CO2(g)����H����283.0 kJ��mol-1
O2(g) ��CO2(g)����H����283.0 kJ��mol-1
C(ʯī)��O2(g) ��CO2(g)����H����393.5 kJ��mol-1
��4Fe(s)��3O2(g) ��2Fe2O3(s)����H�� ��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ̼�ǻ�������������Ԫ�أ��䵥�ʼ������������������������Ҫ��Դ���ʣ�
̼�ǻ�������������Ԫ�أ��䵥�ʼ������������������������Ҫ��Դ���ʣ��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ���¿α���������¿���������ѧ�Ծ���B�����������棩 ���ͣ������
��8�֣�̼�ǻ�������������Ԫ�أ��䵥�ʼ������������������������Ҫ��Դ���ʡ�
��1��ʹCl2��H2O(g)ͨ�����ȵ�̿�㣬����HCl��CO2������1molCl2���뷴Ӧʱ�ͷų�145kJ������д���÷�Ӧ���Ȼ�ѧ����ʽ�� ��
��2����ѧ������̫���ֽܷ�ˮ���ɵ������ڴ����������������̼��Ӧ���ɼ״�����֪H2(g)��CO(g)��CH3OH(l)��ȼ���Ȧ�H�ֱ�Ϊ��285.8kJ��mol-1����283.0kJ��mol-1�ͣ�726.5kJ��mol-1����
����̫���ֽܷ�10molˮ���ĵ�������_____________kJ��
�ڼ״�����ȫȼ������һ����̼��Һ̬ˮ���Ȼ�ѧ����ʽΪ_______ ____ ��
��3����֪��Fe2O3(s)��3C(ʯī)��2Fe(s)��3CO(g) ��H����489.0 kJ��mol-1
CO(g)��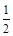 O2(g) ��CO2(g)����H����283.0 kJ��mol-1
O2(g) ��CO2(g)����H����283.0 kJ��mol-1
C(ʯī)��O2(g) ��CO2(g)����H����393.5 kJ��mol-1
��4Fe(s)��3O2(g) ��2Fe2O3(s)����H�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ������ʡ��������УЭ����߶����ϣ����л�ѧ�Ծ��������棩 ���ͣ������
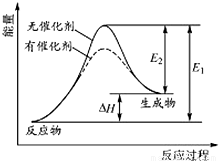
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com