
| A����ˮ�м�ˮϡ�ͺ���Һ�г���H2O����������Ũ�ȶ��½� |
| B��25��ʱ����10mL pH=a��������100mLpH=b��Ba��OH��2��Һ��Ϻ�ǡ���кͣ���a+b=13 |
| C�������ʵ���Ũ�ȵ�NaCl��Һ��CH3COONa��Һ�������ϣ�������Һ������Ũ�ȴ�С��ϵΪ��c��C1-����c��Na+����c��CH3COO-����c��OH-����c��H+�� |
| D������������ʵ���Ũ�ȵ�NaX������HX��Ϻ����Һ�Լ��ԣ�������Һ����Ũ�ȴ�С��ϵΪ��c��X-����c��Na+����c��HX����c��OH-����c��H+�� |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����Һ��pHֵ��С |
| B������ĵ�������� |
| C����Һ�ĵ���������ǿ |
| D���кʹ���Һ�����0.1mol?L-1 NaOH��Һ��������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| c(OH-) |
| c(H+) |
| A����һԪ����Һ��pH=1 | ||
| B������Һ��HA�ĵ����Ϊ1% | ||
| C������Һ��ˮ�����ӻ�����Ϊ1��10-22 | ||
D������ˮϡ�ͣ���
|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ��ʴ���
| c(OH-) |
| c(NH3?H2O) |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ��ʴ���
| c(CH3COO-)?c(H+) |
| c(CH3COOH) |
| c(CH3COOH) |
| c(CH3COO-)c(H+) |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ϡ��Һ��ˮ�����ӻ������Ĵ�С���¶ȡ�Ũ���й� |
| B��Ka��HCN����Kb��CH3COOH��˵����ͬ���ʵ���Ũ��ʱ������������Աȴ���ǿ |
| C������������Һ�е���ͬŨ�ȵ��Ȼ��ƺ͵⻯����Һ���Ȳ���AgI��������Ksp��AgI����Ksp��AgCl�� |
| D�����¶�����ʱ��ˮ�����ӻ�������KW����������볣����Kb������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
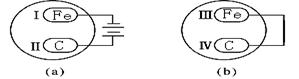
| A����͢� | B����͢����� | C����͢� | D����͢����� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com