
�ζ�ʵ���ǻ�ѧѧ������Ҫ�Ķ���ʵ�顣
��ش��������⣺
��1������к͵ζ������ñ�����ζ�δ֪Ũ�ȵ�NaOH��Һ��
�����в�����ɲⶨ���ƫ�ߵ��� (��ѡ����ĸ)
A���ζ��յ����ʱ�����ӵζ��̶ܿȣ�����������ȷ��
B��ʢװδ֪Һ����ƿ������ˮϴ����δ��δ֪Һ��ϴ
C����ʽ�ζ���������ˮϴ����δ�ñ�������ϴ
D���ζ�ǰ���ζ��ܼ��������ݣ��ζ���������ʧ
�ڸ�ѧ����ʵ��������£�
A���ü�ʽ�ζ���ȡϡNaOH 25.00mL��ע����ƿ�У����������ָʾ����
B���ô��ⶨ����Һ��ϴ��ʽ�ζ��ܡ�
C��������ˮϴ�ɾ��ζ��ܡ�
D��ȡ����ʽ�ζ����ñ���HCl��Һ��ϴ����Һע��ζ��̶ܿȡ�0������2��3cm�����ٰѵζ��̶ܹ��ã�����Һ�����̶ȡ�0����0���̶����¡�
E�����ζ����Ƿ�©ˮ��
F����ȡ��ƿ�����ظ�����һ�Ρ�
G������ƿ���ڵζ������棬ƿ�µ�һ�Ű�ֽ���ߵα�ҡ����ƿֱ���ζ��յ㣬���µζ���Һ�����ڿ̶ȡ�
a. �ζ���������ȷ˳���ǣ��������д��___________________________ ��
b. ��G���������ȷ���յ㣿 __________________________ ____ ��
c. ������̪��ָʾ�������ȷ���յ㣿 _________________________ ____ ��
��2��������ԭ�ζ���ȡ������Һ������ƿ�У���������ϡ���ᣬ��Ũ��Ϊ0.1mol��L��1�ĸ��������Һ�ζ��������ķ�ӦΪ��2KMnO4+5H2C2O4+3H2SO4=K2SO4+10CO2��+2MnSO4+8H2O�������м�¼��ʵ�����ݣ�
|
����� |
����Һ��� (mL)[�� |
��KMnO4��Һ���(mL) |
|
|
�ζ�ǰ���� |
������ |
||
|
��һ�� |
25.00 |
0.50 |
20.40 |
|
�ڶ��� |
25.00 |
3.00 |
23.00 |
|
������ |
25.00 |
4.00 |
24.10 |
�ٵζ�ʱ��KMnO4��ҺӦװ�� (���ᡱ�)ʽ�ζ����У��ζ��յ�ʱ�ζ������� ��
�ڸò�����Һ�����ʵ���Ũ��Ϊ_____________��
��3�������ζ��D�D�ζ����ͱ��ζ����������ȵζ�����ָʾ��������������ܡ�
�ο��±��е����ݣ�����AgNO3�ζ�NaSCN��Һ����ѡ�õ�ָʾ���� (��ѡ����ĸ)��
|
������ |
AgCl |
AgBr |
AgCN |
Ag2CrO4 |
AgSCN |
|
��ɫ |
�� |
dz�� |
�� |
ש�� |
�� |
|
Ksp |
1.77��10��10 |
5.35��10��13 |
1.21��10��16 |
1.12��10��12 |
1.0��10��12 |
A��NaCl B��NaBr C��NaCN D��Na2CrO4
��16�֣���1����CD ����a.ECBADGF��ECDBAGF��b.��Һ�ɻ�ɫ���ɫ�Ұ�����ڲ��ָ���c.��Һ��dz��ɫ����ɫ�Ұ�����ڲ��ָ�
��2�������ƿ����Һ����ɫ��Ϊ��ɫ���Ұ�����ڲ���ɫ����0.2 mol��L��1
��3��D
��������
�����������1��A���ζ��յ����ʱ���Ӷ������V������ƫС������c�����⣩=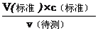
��������֪c������ƫС����A����B����ƿˮϴ������δ֪Һϴ�ӣ��Խ����Ӱ�죬����C����ʽ�ζ���ʹ��ǰ��ˮϴ��δ�ñ�������ϴ�����������ʵ���Ũ��ƫС�����V������ƫ����c�����⣩= ��������֪c�����⣩ƫ��C��ȷ��D����ʽ�ζ��ܼ��첿�������ݣ��ζ�����ʧ�����V������ƫ����c�����⣩=
��������֪c�����⣩ƫ��C��ȷ��D����ʽ�ζ��ܼ��첿�������ݣ��ζ�����ʧ�����V������ƫ����c�����⣩= ��������֪c������ƫ��D��ȷ����2������ΪKMnO4����ǿ�����ԣ��ḯʴ�ܣ���Ӧ����ʽ�ζ���ʢװ����KMnO4��Һ��������ɫ��Ϊָʾ���жϵζ��յ�ʱ���ٵμ�KMnO4��Һʱ����Һ������ɫ��Ϊ��ɫ����������Ũ��Ϊc��
��������֪c������ƫ��D��ȷ����2������ΪKMnO4����ǿ�����ԣ��ḯʴ�ܣ���Ӧ����ʽ�ζ���ʢװ����KMnO4��Һ��������ɫ��Ϊָʾ���жϵζ��յ�ʱ���ٵμ�KMnO4��Һʱ����Һ������ɫ��Ϊ��ɫ����������Ũ��Ϊc��
2KMnO4 + 5H2C2O4
2 5
20��10-3 L��0.1mol��L��1 25��10-3 L��c
���c=0.2 mol��L��1
��3������AgNO3ȥ�ζ�NaSCN��Һ����ѡ�õĵζ�ָʾ�������ʵ��ܽ��Ӧ��AgSCN�����������ԣ�ӦΪNa2CrO4��
���㣺�ζ�ʵ�顢������ �����ܵ���ʵ��ܽ�ƽ�⼰����ת���ı��ʡ���ѧ����ʽ���йؼ��㡣



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| �ζ����� | ����Һ��� ��mL�� |
��KMnO4��Һ�����mL�� | |
| �ζ�ǰ���� | �ζ������ | ||
| ��һ�� | 25.00 | 0.50 | 20.40 |
| �ڶ��� | 25.00 | 3.00 | 23.00 |
| ������ | 25.00 | 4.00 | 24.10 |
| ������ | AgCl | AgBr | AgCN | Ag2CrO4 | AgSCN |
| ��ɫ | �� | dz�� | �� | ש�� | �� |
| Ksp | 1.77��10-10 | 5.35��10-13 | 1.21��10-16 | 1.12��10-12 | 1.0��10-12 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�����ʡƽ��ɽ�и߶���ѧ����ĩ���Ի�ѧ�Ծ��������棩 ���ͣ�ʵ����
�ζ�ʵ���ǻ�ѧѧ������Ҫ�Ķ���ʵ�顣
��ش��������⣺
��1������к͵ζ������ñ�����ζ�δ֪Ũ�ȵ�NaOH��Һ�����в�����ɲⶨ���ƫ�ߵ��� (��ѡ����ĸ)
A���ζ��յ����ʱ�����ӵζ��̶ܿȣ�����������ȷ��
B��ʢװδ֪Һ����ƿ������ˮϴ����δ��δ֪Һ��ϴ
C����ʽ�ζ���������ˮϴ����δ�ñ�������ϴ
D���ζ�ǰ���ζ��ܼ��������ݣ��ζ���������ʧ
��2��������ԭ�ζ�����ȡ������Һ������ƿ�У���������ϡ���ᣬ��Ũ��Ϊ0.1mol��L��1�ĸ��������Һ�ζ��������ķ�ӦΪ��2KMnO4+5H2C2O4+3H2SO4=K2SO4+10CO2��+2MnSO4+8H2O�������м�¼��ʵ�����ݣ�
|
����� |
����Һ��� (mL) |
��KMnO4��Һ���(mL) |
|
|
�ζ�ǰ���� |
������ |
||
|
��һ�� |
25.00 |
0.50 |
20.40 |
|
�ڶ��� |
25.00 |
3.00 |
23.00 |
|
������ |
25.00 |
4.00 |
24.10 |
�ٵζ�ʱ��KMnO4��ҺӦװ�� (��ᡱ�)ʽ�ζ����У��ζ��յ�ʱ�ζ�������
�ڸò�����Һ�����ʵ���Ũ��Ϊ_____________��
��3�������ζ��D�D�ζ����ͱ��ζ����������ȵζ�����ָʾ��������������ܡ�
�ο��±��е����ݣ�����AgNO3�ζ�NaSCN��Һ����ѡ�õ�ָʾ���� (��ѡ����ĸ)��
|
������ |
AgCl |
AgBr |
AgCN |
Ag2CrO4 |
AgSCN |
|
��ɫ |
�� |
dz�� |
�� |
ש�� |
�� |
|
Ksp |
1.77��10��10 |
5.35��10��13 |
1.21��10��16 |
1.12��10��12 |
1.0��10��12 |
A��NaCl B��NaBr C��NaCN D��Na2CrO4
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ��ɽ��ʡ�����в�ɽ�ظ�����ѧ����ĩ��⻯ѧ�Ծ� ���ͣ�ʵ����
��8�֣���1����ѧ��һ����ʵ��Ϊ������ѧ�ƣ���ѧʵ���ǻ�ѧѧϰ����Ҫ���ݡ����������յ�֪ʶ�жϣ�����ʵ������������У���ȷ���� ������ĸ��ţ���
A�����Լ�ƿ��ȡ����ҩƷ������ʣ���ٷŻ�ԭ�Լ�ƿ
B������һ��������ͬ����ֽ����ƽ���������ϣ���NaOH�����������ֽ�ϳ���
C���������ʱ�����ܽ���ˮ�ķ������Ͽڽ����¿ڳ�
D��NaOH��Һ���ܱ����ڴ��в��������Լ�ƿ��
E����ϡ����ε�pH��ֽ�ϣ��������ɫ���Ƚϣ��ⶨϡ�����pH
F����Һ������ʾ����Һ©�����²�Һ����¿ڷų����ϲ�Һ���Ͽڵ���
��2����ѧʵ���У�����ȷ�IJ������ʵ������ȷ�����һ����Ӱ�죬���á�>������<����=����д���пհף�
����������ƽ��ȡ8.4g�Ȼ��ƣ�����������Ȼ��Ƶ�λ�÷ŵߵ��ˣ�����ȡ�Ȼ��Ƶ�����
8.4g��
��������ƿ����500mL0.1mol��L-1NaOH��Һ������ʱ���ӿ̶��ߣ�����NaOH��Һ�����ʵ���Ũ�� 0.1mol��L-1��
���к͵ζ�ʵ��ʱ��ƿ������ˮϴ����δ�ñ�Һ��ϴ��������ƿ�д���Һ��Ũ�� ʵ��Ũ�ȡ�
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com