
(1)д���Ҵ���ȫȼ�յĻ�ѧ����ʽ��______________________________________________��
(2)�Ҵ�ȼ��ʱ����������㣬���ܻ���CO���ɡ�����ͼװ����֤�Ҵ���ȼ�ղ�������CO��CO2��H2O��Ӧ���Ҵ���ȼ�ղ�������ͨ��(�����������ҵ�˳����װ�ñ��)__________��
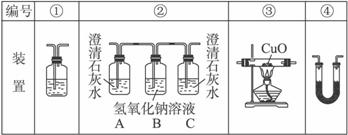
(3)ʵ��ʱ�ɹ۲쵽װ�â���Aƿ��ʯ��ˮ����ǡ�
Aƿ��Һ��������_______________________________________________________��
Bƿ��Һ��������_______________________________________________________��
Cƿ��Һ��������_______________________________________________________��
(4)װ�â۵�������____________________________��װ�â�����ʢ�Լ���______________________��Һ��������______________________________________��
(5)װ�â�����ʢ�Ĺ���ҩƷ��_________________����������֤�IJ�����_______________��
(6)β���Ĵ���������______________________________________________________��
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ѵ����ѧ�ս̰� �ս̰� ���ͣ�058
����ȼ���Ҵ���ҵ��ʮһ�塱�滮����2010�꣬�Ҵ����ͽ�ռ�й�������������һ�����ϣ��Ҵ�������Ϊһ���������ȼ�ϣ������ҹ��Ĺ��ұ����Ҵ���������90%����ͨ������10%��ȼ���Ҵ����Ͷ��ɣ�����Ӱ����������ʻ���ܣ��������к�������ŷ�����
(1)д���Ҵ���ȫȼ�յĻ�ѧ����ʽ��________��
(2)�Ҵ�ȼ��ʱ����������㣬���ܻ���CO���ɣ�����ͼװ����֤�Ҵ���ȼ�ղ�������CO��CO2��H2O��Ӧ���Ҵ���ȼ�ղ�������ͨ��(�����������ҵ�˳����װ�ñ��)________��
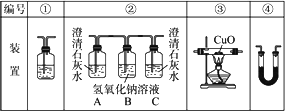
(3)ʵ��ʱ�ɹ۲쵽װ�â���Aƿ��ʯ��ˮ����ǣ�
Aƿ��Һ��������________��
Bƿ��Һ��������________��
Cƿ��Һ��������________��
(4)װ�â۵�������________��װ�â�����ʢ�Լ���________��Һ��������________��
(5)װ�â�����ʢ�Ĺ���ҩƷ��________����������֤�IJ�����________��
(6)β���Ĵ���������________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013����ʡ����һ�и߶���ѧ�����п��Ի�ѧ�Ծ����������� ���ͣ������
����ʮ�ߴ�ָ��������ǿ��Դ��Դ��Լ����̬������������ǿ�ɳ�����չ��������ֽ�Լ��Դ�ͱ��������Ļ������ߣ���չ������ҵ����
(1)�����й���������������Ҫ�����________��
| A����úҺ�������������ȼ�ϵ�ȼ��Ч�� |
B����װ����β����ת��װ�ã�ʹ֮��Ӧ��4CO��2NO2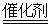 4CO2��N2 4CO2��N2 |
| C�������ƹ��Ҵ����͵�ͬʱ���о�����̫������������ȼ�յ������ |
| D����ˮ�����硢�������硢���ܷ���ͷ���������Ҫ������չ�������� |
 CO(g)��H2(g)��C(s)��H2(g)��CO(g)��ȫȼ�յ��Ȼ�ѧ����ʽΪ��
CO(g)��H2(g)��C(s)��H2(g)��CO(g)��ȫȼ�յ��Ȼ�ѧ����ʽΪ��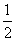 O2(g)===H2O(g) ��H����242.0 kJ/mol ��
O2(g)===H2O(g) ��H����242.0 kJ/mol ��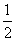 O2(g)===CO2(g) ��H����283.0 kJ/mol�� ��
O2(g)===CO2(g) ��H����283.0 kJ/mol�� ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013����ʡ�߶���ѧ�����п��Ի�ѧ�Ծ��������棩 ���ͣ������
����ʮ�ߴ�ָ��������ǿ��Դ��Դ��Լ����̬������������ǿ�ɳ�����չ��������ֽ�Լ��Դ�ͱ��������Ļ������ߣ���չ������ҵ����
(1)�����й���������������Ҫ�����________��
A����úҺ�������������ȼ�ϵ�ȼ��Ч��
B����װ����β����ת��װ�ã�ʹ֮��Ӧ��4CO��2NO2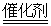 4CO2��N2
4CO2��N2
C�������ƹ��Ҵ����͵�ͬʱ���о�����̫������������ȼ�յ������
D����ˮ�����硢�������硢���ܷ���ͷ���������Ҫ������չ��������
(2)��úת��Ϊˮú������Ҫ��ѧ��ӦΪ
C(s)��H2O(g) CO(g)��H2(g)��C(s)��H2(g)��CO(g)��ȫȼ�յ��Ȼ�ѧ����ʽΪ��
CO(g)��H2(g)��C(s)��H2(g)��CO(g)��ȫȼ�յ��Ȼ�ѧ����ʽΪ��
C(s)��O2(g)===CO2(g) ��H����393.5 kJ/mol�� ��
H2(g)��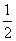 O2(g)===H2O(g) ��H����242.0 kJ/mol ��
O2(g)===H2O(g) ��H����242.0 kJ/mol ��
CO(g)��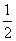 O2(g)===CO2(g)
��H����283.0 kJ/mol��
��
O2(g)===CO2(g)
��H����283.0 kJ/mol��
��
��ش�
�ٸ����������ݣ�д��C(s)��H2O(g)��Ӧ���Ȼ�ѧ����ʽ��_____________________________________.
��ˮú������������������ȼ�ϣ�Ҳ����Ҫ���л�����ԭ�ϣ�CO��H2��һ�������¿��Ժϳɣ�a.�״���b.��ȩ��c.���d.���ᣮ�Է�����CO��H2��1��1������Ȼ�Ϸ�Ӧ���ϳ�����________(�����)����ʱ���������㡰��ɫ��ѧ����Ҫ����ȫ����ԭ���е�ԭ�ӣ�ʵ�����ŷţ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��1��д���Ҵ���ȫȼ�յĻ�ѧ����ʽ��___________________________________________��
��2���Ҵ�ȼ��ʱ����������㣬���ܻ���CO���ɡ�����ͼװ����֤�Ҵ���ȼ�ղ�������CO��CO2��H2O��Ӧ���Ҵ���ȼ�ղ�������ͨ�����������������ҵ�˳����װ�ñ��_________��
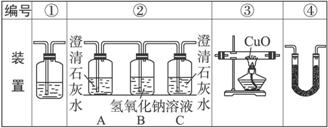
��3��ʵ��ʱ�ɹ۲쵽װ�â���Aƿ��ʯ��ˮ����ǡ�Aƿ��Һ��������________________��Bƿ��Һ��������______________________��Cƿ��Һ��������______________________��
��4��װ�â۵�������___________________��װ�â�����ʢ�Լ���___________��Һ��������_________________________________��
��5��װ�â�����ʢ�Ĺ���ҩƷ��___________����������֤�IJ�����___________��
��6��β���Ĵ���������___________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com