
��14�֣���ѧ��ѧ����������A��B��C��D֮���������ת����ϵ��
A + B �� C + D + H2O���밴Ҫ����գ�
��1����AΪ������Ԫ����ɵĺ�ɫ���嵥�ʣ���B��Ũ��Һ����ʱ������C��D��������,
C��D�����������ʹ����ʯ��ˮ����ǣ���A��B��Ӧ�ķ���ʽ�� ����֪���д̼�����ζ��C�����Cl2����Ư��ijЩ��ɫ���ʣ������������������ʵ���ͨ�뵽Ʒ����Һ�е�ʵ������Ϊ ԭ���� ��
�������������ǵ���ͨ�뵽ˮ�У�Ϊ����֤�������������ij��ȤС���ͬѧ���������Լ��� �� �Ȼ�����Һ �� �Ȼ�������Һ �� ���軯����Һ �� ������Һ
�� Ʒ����Һ �� ���Ը��������Һ
��Cl2������ȡ������Һ�μ���ʢ�� ��ѡ��һ����ţ��Լ����Թ��ڣ��ټ���
��ѡ��һ����ţ��Լ��������������ǣ� ��
��C������ȡ������Һ�μ���ʢ�� ��ѡ��һ����ţ��Լ����Թ��ڣ�����������
�ǣ� ��
��2����A��ˮ�е��ܽ�����¶ȵ����߶����ͣ�BΪ�����ڷǽ������ʣ�C��Ư�۵���Ч�ɷ֣���C����ˮ�ⷴӦ�����ӷ���ʽ�� ��
��3����AΪ���10���ӵ��������뵥��18���ӵ������ӹ��ɵ���ɫ���壬�����ֽ⣬�ֽ���������ּ�������ˮ�����塣����A�������ӵķ�����
�������������̼����ۣ���
��1��C + 2H2SO4(Ũ) 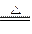 CO2��+2SO2��+2H2O ��2�֣�
CO2��+2SO2��+2H2O ��2�֣�
������1�֣���Cl2+SO2+2H2O��4H++2Cl�D+SO42�D��ʧȥƯ���ԣ�2�֣�
�ڣ�1�֣� �ۣ���ܣ���1�֣� ��Һ���ֺ�ɫ������ɫ����1�֣�
�ݣ���ޣ���1�֣� ��ɫ��ȥ���Ϻ�ɫ��ȥ����1�֣�
��2��ClO��+ H2O  HClO + OH�� ��2�֣�
HClO + OH�� ��2�֣�
��3��ȡ����A����ˮ�����Һ���Թ��У������Һ�м���NaOH�����ȣ���ʪ��ĺ�ɫʯ����ֽ���ڹܿڼ����������壬ʯ����ֽ����֤����Һ�к���NH4+�� ������Ũ������飬�а������ɣ� ��2�֣�
����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��14�֣���ѧ��ѧ����������A��B��C��D֮���������ת����ϵ��
A + B �� C + D + H2O���밴Ҫ����գ�
��1����AΪ������Ԫ����ɵĺ�ɫ���嵥�ʣ���B��Ũ��Һ����ʱ������C��D��������,
C��D�����������ʹ����ʯ��ˮ����ǣ���A��B��Ӧ�ķ���ʽ�� ����֪���д̼�����ζ��C�����Cl2����Ư��ijЩ��ɫ���ʣ������������������ʵ���ͨ�뵽Ʒ����Һ�е�ʵ������Ϊ ԭ���� ��
�������������ǵ���ͨ�뵽ˮ�У�Ϊ����֤�������������ij��ȤС���ͬѧ���������Լ��� �� �Ȼ�����Һ �� �Ȼ�������Һ �� ���軯����Һ �� ������Һ
�� Ʒ����Һ �� ���Ը��������Һ
��Cl2������ȡ������Һ�μ���ʢ�� ��ѡ��һ����ţ��Լ����Թ��ڣ��ټ���
��ѡ��һ����ţ��Լ��������������ǣ� ��
��C������ȡ������Һ�μ���ʢ�� ��ѡ��һ����ţ��Լ����Թ��ڣ�����������
�ǣ� ��
��2����A��ˮ�е��ܽ�����¶ȵ����߶����ͣ�BΪ�����ڷǽ������ʣ�C��Ư�۵���Ч�ɷ֣���C����ˮ�ⷴӦ�����ӷ���ʽ�� ��
��3����AΪ���10���ӵ��������뵥��18���ӵ������ӹ��ɵ���ɫ���壬�����ֽ⣬�ֽ���������ּ�������ˮ�����塣����A�������ӵķ�����
�������������̼����ۣ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�������ʡ�����и���9���¿���ѧ���� ���ͣ������
��12�֣���ѧ��ѧ����������A��B��C��D֮���������ת����ϵ��
A + B �� C + D + H2O���밴Ҫ����գ�
��1����AΪ������Ԫ����ɵĺ�ɫ���嵥�ʣ���B��Ũ��Һ����ʱ������C��D�������塣C��D�����������ʹ����ʯ��ˮ����ǣ���÷�Ӧ�Ļ�ѧ����ʽ
�� ��
�������������岻��ѡ�õ��Լ��� ��
a��BaCl2��Һ�� b��KMnO4��Һ c��Ʒ����Һ������d���ữ��Ba��NO3��2��Һ
��500mL 2mol/L��NaOH ��Һ��ͨ��0.8mol��ɫ��ζ��C���壬ǡ����ȫ��Ӧ���÷�Ӧ���ܻ�ѧ����ʽ�� ����ʱ��Һ�е����Ӱ���Ũ���ɴ�С���е�˳���� ��
��2����A��ˮ�е��ܽ�����¶ȵ����߶����ͣ�BΪ�����ڷǽ������ʣ�D��Ư�۵ijɷ�֮һ��C����ˮ�ⷴӦ�����ӷ���ʽ�� ��
��3����AΪ���10���ӵ��������뵥��18���ӵ������ӹ��ɵ���ɫ���壬�����ֽ⣬�ֽ���������ּ�������ˮ�����塣����A�������ӵķ�����
�������������̼����ۣ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�������ʡ����9���¿���ѧ���� ���ͣ������
��12�֣���ѧ��ѧ����������A��B��C��D֮���������ת����ϵ��
A + B �� C + D + H2O���밴Ҫ����գ�
��1����AΪ������Ԫ����ɵĺ�ɫ���嵥�ʣ���B��Ũ��Һ����ʱ������C��D�������塣C��D�����������ʹ����ʯ��ˮ����ǣ���÷�Ӧ�Ļ�ѧ����ʽ
�� ��
�������������岻��ѡ�õ��Լ��� ��
a��BaCl2��Һ�� b��KMnO4��Һ c��Ʒ����Һ������d���ữ��Ba��NO3��2��Һ
��500mL 2mol/L��NaOH ��Һ��ͨ��0.8mol��ɫ��ζ��C���壬ǡ����ȫ��Ӧ���÷�Ӧ���ܻ�ѧ����ʽ�� ����ʱ��Һ�е����Ӱ���Ũ���ɴ�С���е�˳���� ��
��2����A��ˮ�е��ܽ�����¶ȵ����߶����ͣ�BΪ�����ڷǽ������ʣ�D��Ư�۵ijɷ�֮һ��C����ˮ�ⷴӦ�����ӷ���ʽ�� ��
��3����AΪ���10���ӵ��������뵥��18���ӵ������ӹ��ɵ���ɫ���壬�����ֽ⣬�ֽ���������ּ�������ˮ�����塣����A�������ӵķ�����
�������������̼����ۣ���
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com