
�л���A��B��C��D��E��F��G�����ϵ����ͼ��ʾ��
��1������A��±Ԫ�ص�ʵ�鷽���� ��
��2��B�Ľṹ��ʽΪ ���ٵĻ�ѧ��Ӧ������ ��
��3��G��ʵ�����п�ͨ��������Ӧ������F�����е�һ����Ӧ�������� ����Ӧ�õ��IJ�������У���д�ṹ��ʽ�� ��
��4��F��һ�ֶ�Ԫ�ᣬ����һ�������¿���G��Ӧ���ɸ߷��ӻ�����ø߷��ӵĽṹ��ʽΪ ��
д����Ӧ�ܵĻ�ѧ��Ӧ����ʽ ��
��1��ȡ����A���Թܣ���������������Һ����һ��ʱ�䣬��ȴ������ϡ��������Һ��������������������Һ������е���ɫ�������֣���A�к���Ԫ�ء���2�֣�
��2��H2C=CH-COOH��1�֣��� ��ȥ��������Ӧ��1�֣�
��3��Cu��Ag�����������ȣ�1�֣��� HO-CH2-CH2-CHO��OHC-CH2-CHO��2�֣�
��4��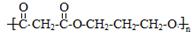 ��1�֣�
��1�֣� +2NaOH
+2NaOH CH2=CHCOONa+HO-CH2-CH2-CH2-OH+NaBr��2�֣���Ӧ�������ȷ1�֣���ƽ+����1�֣�
CH2=CHCOONa+HO-CH2-CH2-CH2-OH+NaBr��2�֣���Ӧ�������ȷ1�֣���ƽ+����1�֣�
���������������1��±������±�����ӵļ��鷽���ǣ�ȡ����A���Թܣ���������������Һ����һ��ʱ�䣬��ȴ�����ϡ��������Һ�����ԣ��μ���������Һ������е���ɫ�������֣���A�к���Ԫ�ء�����A��������������������������ʣ���֪�Ƿ�����ˮ�ⷴӦ��D��B���ữ��˵��B���ᣬ��C�Ǵ�����̼ԭ������̼�ĹǼ���ȫ��ͬ������ת����ϵ��֪BΪH2C=CH-COOH��F��һ�ֶ�Ԫ�ᣬͨ��ת����ϵ�ɵó�CΪBrH2C-CH2-CH2OH��EΪBrH2C-CH2-CHO��DΪH2C=CH-COONa��GΪHOH2C-CH2-CH2OH,FΪHOOC-CH2-COOH��
��2��B�Ľṹ��ʽΪH2C=CH-COOH���ٵĻ�ѧ��Ӧ��������ȥ��������Ӧ��G��ʵ�����п�ͨ��������Ӧ������F�����е�һ����Ӧ��������Cu��Ag�����������ȣ���Ӧ�õ��IJ����в���������ȫ���������ʲ��������HO-CH2-CH2-CHO��OHC-CH2-CHO��
��4��F��һ�ֶ�Ԫ�ᣬ����һ�������¿���G��Ӧ���ɸ߷��ӻ�����൱�ڷ�����������Ӧ�����˸߾��д����Ӧ��Ϊ����±������ˮ�ⷴӦ�������������ŷ����˱仯�����㣺���⿼�����л���ͼ�ƶϣ���һ���ۺ��Ե�ǰ���ƶϣ��ѶȽϴ�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������F(ƥ����͡)���ڸߵ��̴�Ѫ֢�����ƣ���ϳ�·�����£�
(1)������D�й����ŵ�����Ϊ________��________��������
(2)A��B�ķ�Ӧ������____________��
(3)д��ͬʱ��������������A��һ��ͬ���칹��Ľṹ��ʽ��________________��
�ٷ����к��������������ڷ�������3�ֲ�ͬ��ѧ�������⣻�۲�����O��O����
(4)ʵ��D��E��ת���У�������X�ķ���ʽΪC19H15NFBr��д����ṹ��ʽ��________________��
(5)��֪��������E��CF3COOH����������ת��Ϊ ����ת��ΪF������Ϊ�ϳ�·������Ʋ���ڵ�Ŀ����___________________________________________________��
����ת��ΪF������Ϊ�ϳ�·������Ʋ���ڵ�Ŀ����___________________________________________________��
(6)�����ϳ�·���У�����۵IJ����D����� ���÷�Ӧԭ�����л��ϳ��о��й㷺Ӧ�á���д����
���÷�Ӧԭ�����л��ϳ��о��й㷺Ӧ�á���д���� Ϊ��Ҫԭ���Ʊ�
Ϊ��Ҫԭ���Ʊ� �ĺϳ�·������ͼ(���Լ�����)���ϳ�·������ͼʾ�����£�
�ĺϳ�·������ͼ(���Լ�����)���ϳ�·������ͼʾ�����£�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ijѧϰС��Ϊ̽��ά����C����ɺ��й����ʣ�����������ʵ�飺
��ȡά����C��Ʒ���飬����ȡ������0.704 g�����ڲ��۲�����ȼ�չ��У�����ͨ�����������þƾ���Ƴ���������Ʒ����Ʒ����ʧ������κβ������������(������������)�Ⱥ�ͨ����ˮ����ͭ�ͼ�ʯ�ң����߷ֱ�����0.288 g��1.056 g����������ȫ�����ա�
�ڽ����������ά����C��Ʒ����������ɫʯ����Һ��ֻ�Ϻ���Һ��ɫ��졣
�����������⣺
��1����ȡ�������У�������ԭ�ӵ����ʵ����� mo1��̼Ԫ�ص������� g��
��2��ά����C�� (�������������������ȷ���Ƿ�)����Ԫ�أ������Ǹ���ʵ�����ݿ�֪ ��
����ش𡰺����У�������ȡ����������ԭ�ӵ����ʵ�����������һ�ո��У�����ش𡰲������ȣ�����һ�ո���д����ȡ��������ԭ�ӵ����ʵ���Ϊ mol��
��3�����Ҫȷ��ά����C�ķ���ʽ������Ϊ����Ҫ֪������Ϣ�� ��
��4��ά����C����ɫʯ����Һ��Ϻ���Һ��ɫ��죬˵��ά����C��Һ���� (��ᡱ��������С�)�ԡ������й�������ʾ��ά����C���л�ԭ�ԡ����Ե�����Һ����ˮΪ�Լ���д����֤ά����C���л�ԭ�Ե�ʵ�鷽�������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�㶹������;�㷺�����ϣ��ϳ��㶹�ص�·�����£������Լ������P��Ӧ������ʡ�ԣ���
��1����ķ���ʽΪ ��
��2����Ӧ�ڵķ�Ӧ������ ����Ӧ�ܵķ�Ӧ������ ��
��3���㶹���ڹ���NaOH��Һ����ȫˮ��Ļ�ѧ����ʽΪ ��
��4�����Ǣ���ͬ���칹�壬���ķ����к��б�������̼̼˫���������Ϻ���������λȡ�������ܷ���������Ӧ�����Ľṹ��ʽΪ ����дһ�֣���
��5��һ�������£� ��CH3CHO�ܷ������Ʒ�Ӧ�١��ڵ�������Ӧ���������ɵ��л���Ľṹ��ʽΪ ��
��CH3CHO�ܷ������Ʒ�Ӧ�١��ڵ�������Ӧ���������ɵ��л���Ľṹ��ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��A�����������ʵ���1:1�����ӳɷ�Ӧʱ���Եõ����ֲ���������������ֲ���Ľṹ��ʽ��
| ���� | �� | �� |
| �ṹ��ʽ | 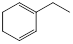 | 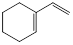 |
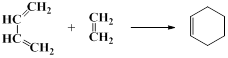
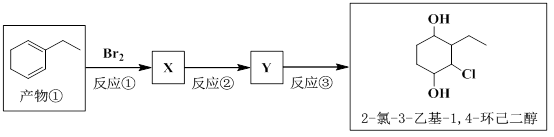
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��Է�������Ϊ92��ij������X��һ����Ҫ���л�����ԭ�ϣ��о���������Ϊ��ʼԭ����Ƴ�����ת����ϵͼ�����ֲ���ϳ�·�ߡ���Ӧ������ȥ��������A��һ�ȴ��H��һ�ֹ��ܸ߷��ӣ��������ΪC7H5NO��
��֪��
��
��
�������ѧ֪ʶ�뱾��������Ϣ�ش��������⣺
��1��H�Ľṹ��ʽ�ǣ� ��X��A�ķ�Ӧ���� ��
��2����Ӧ�ڵ������� ����Ӧ�ں͢��Ⱥ�˳���ܵߵ���ԭ���� ��
��3��д����Ӧ�ٵĻ�ѧ����ʽ�� ��
��4�� �ж���ͬ���칹�壬д��2�ֺ���1��ȩ����2���ǻ��ұ�����ֻ��2��һ��ȡ����ķ����廯����Ľṹ��ʽ�� ��
�ж���ͬ���칹�壬д��2�ֺ���1��ȩ����2���ǻ��ұ�����ֻ��2��һ��ȡ����ķ����廯����Ľṹ��ʽ�� ��
��5��д����Aת��Ϊ ��·�ߡ�����A�� ���� ��
��·�ߡ�����A�� ���� �� �ڼ�����д����Ӧ�Լ�����Ӧ��������
�ڼ�����д����Ӧ�Լ�����Ӧ��������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������A��C11H8O4��������������Һ�м��ȷ�Ӧ�����ữ�ɵõ�������B��C���ش��������⣺
��1��B�ķ���ʽΪC2H4O2��������ֻ��һ�������š���B�Ľṹ��ʽ��________��B���Ҵ���Ũ������¼��ȷ�Ӧ����D���÷�Ӧ�Ļ�ѧ����ʽ��________________________���÷�Ӧ��������________��
��2��д�������ܷ���������Ӧ��B��ͬ���칹��Ľṹ��ʽ________________________��
��3��C�Ƿ��㻯�����Է�������180����̼����������Ϊ60.0���������������Ϊ4.4��������Ϊ������C�ķ���ʽ��________��
��4����֪C�ķ�����������ȡ����������һ��ȡ������֧����������ʹ������Ȼ�̼��Һ��ɫ�Ĺ����ż�����̼��������Һ��Ӧ�ų�����Ĺ����ţ����ȡ�����ϵĹ����������� ����������ȡ������ͬ���ֱ�λ�ڸ�ȡ��������λ�Ͷ�λ����C�Ľṹ��ʽ�� ��
��5��A�Ľṹ��ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ϩ��һ����Ҫ�Ļ���ԭ�ϣ�����ϩΪԭ�����������ֻ�����Ʒ�ķ�Ӧ���£����ַ�Ӧ��������ȥ����
��ش��������⣺
��1��д��A��ʵ��������ϩ�ķ���ʽ�� ��
��2��B��E��Ӧ����F�Ļ�ѧ����ʽΪ______ _____���÷�Ӧ������Ϊ_____________��
��3��D�Ľṹ��ʽΪ____ _______��
��4��д��D��ͬ���칹�壨��ȩ��������������Ӧ�Ļ�ѧ����ʽ�� ��
��5��C��ͬ���칹���к�ȩ���Ľṹ�� �֡�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
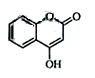 ��һ��ҽҩ�м��壬�������Ʊ�����Ѫҩ����ͨ������·�ߺϳɣ�
��һ��ҽҩ�м��壬�������Ʊ�����Ѫҩ����ͨ������·�ߺϳɣ�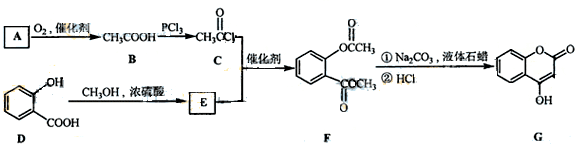
��1��A��������Һ��Ӧ���������ɣ���A�Ľṹ��ʽ�� ��
��2��B��C�ķ�Ӧ������ ��
��3��E�Ľṹ��ʽ��
��4��д��F����NaOH��Һ����ʱ��Ӧ�Ļ�ѧ����ʽ
��5�����й���G��˵����ȷ���� ��
a�������嵥�ʷ�Ӧ b����������Ʒ�Ӧ
c��1molG����ܺ�3mol������Ӧ d������ʽ��C9H6O3
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com