
��15�֣�����������һ�ְ�ɫ�й���Ƭ״�ᾧ���ɫ�ᾧ��ĩ���ǻǰ���ҩ���ԭ�ϣ�������ֹʹ�������ȼ�����������Ⱦ���м��塣
�����������Ʊ�ԭ��Ϊ�� 
ʵ�������
| ���� | ʽ�� | ��״ | �ܶ�/g/cm3 | �е�/�� | �ܽ�� | |
| �� �� | 93 | ��ɫ��״Һ�壬 ���л�ԭ�� | 1.02 | 184.4 | ����ˮ | �������Ҵ������ѵ� |
| �� �� | 60 | ��ɫҺ�� | 1.05 | 118.1 | ������ˮ | �������Ҵ������� |
| �������� | 135 | ��ɫ���� | 1.22 | 304 | ������ˮ��������ˮ | �������Ҵ������� |

��1����ֹ�����ڷ�Ӧ�����б�������
��2�����Ϸֳ���Ӧ���������ɵ�ˮ���ٽ���Ӧ�������
��3���¶ȼ��¶��½�����ƿ��Һ�岻�����ӣ�
��4����ԡ���ò�����Ħ�������ڱڣ���Ͷ�뾧�ֻ��ñ�ˮ��ȴ����
��5��A ����6��60%
���������������1���������ȶ��������ڿ����е���������Ϊ��������Ϊ��ֹ�����ڷ�Ӧ�����б����������뻹ԭ��Zn�ۡ���2��ˮ�ķе���100�棬������105�����ң��Ϳ��Բ��Ϸֳ���Ӧ���������ɵ�ˮ���ٽ���Ӧ������У����������IJ��ʡ���3���������¶ȼ��¶��½�����ƿ��Һ�岻�����Ӿͱ��淴Ӧ����������٣�������ﵽ�����������Ӧ������ɣ���4����4�����ڲ���1���ȵ��¶���105�����ң������ڵ�ˮ�¶�Ϊ100�棬���ܴﵽ���¶ȣ����Բ���1������ԡ���ȵķ���������2�нᾧʱ������ȴ���������������ɲ��õķ������ò�����Ħ�������ڱڣ���Ͷ�뾧�ֻ��ñ�ˮ��ȴ���ķ���ʵ�ֽᾧ����5��������������������ˮ��������ˮ������ϴ�Ӵ�Ʒ����ʵ��Լ�����������ˮϴ���Լ�����ϴ����ɵ���ġ���ѡ��ΪA����6��n(����)=" (9.1mL" ��1.02g/ml)��93g/mol=0.1mol��n(����)=( 17.4mL ��1.05g/ml)��60g/mol=0.305mol�����ڱ��������ʵ��������Բ����������������ʵ���Ҫ�Բ������ı���Ϊԭ��Ϊ�������۲���Ϊ0.1mol����ʵ�ʲ���n(��������)= 8.1g��135g/mol=0.06mol���������������IJ���Ϊ0.06mol��0.1mol��100%=60%��
���㣺�������ʵ��Ʊ��������Ŀ��Ƽ�ʵ���������Ӧ�̶ȵ��жϡ����ʵIJ��ʵļ����֪ʶ��


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����ʵ���ܴﵽԤ��Ŀ�ĵ���
| A����ͼ����֤ͭ��ϡ���ᷴӦ���������ֻ��NO |
| B����ͼ��ʵ������ȡ�������� |
| C����ͼ����֤����Ƭ����������ʴ |
| D����ͼ��֤������ͬ�¶���Mg(OH)2���ܽ�ȴ���Fe(OH)3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
��10�֣���������п��;�dz��㷺��������п��Ʒ�������������CuO��SiO2�����ʣ�Ϊԭ���Ʊ���������п��������������Ʒ�����������������£�
��֪��Zn(OH)2������NaOH��Һ��
��ش��������⣺
��1�����ܽ⡱ǰ������п��Ʒ�����ϸ��������Ŀ���� ��
��2������A������������Ҫ�ɷ��� ��
��3���������Feʱ����ҺM����Fe��Ӧ�������� ��
��4�� ����C���õ�����P�к���Fe(OH)2��Fe(OH)3����д������Fe(OH)3�Ļ�ѧ��Ӧ����ʽΪ ��
��5�����������ijɷ�ΪZnCO3��2Zn(OH)2��H2O���������������ʽ������дΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��15�֣�ijС����CoCl2��6H2O��NH4Cl��H2O2��Ũ��ˮΪԭ�ϣ��ڻ���̿���£��ϳ��˳Ȼ�ɫ����X��Ϊ�ⶨ����ɣ���������ʵ�顣
�ٰ��IJⶨ����ȷ��ȡwgX��������ˮ�ܽ⣬ע����ͼ��ʾ������ƿ�У�Ȼ����μ�������10%NaOH��Һ��ͨ��ˮ����������ƷҺ�еİ�ȫ����������V1mLc1 mol��L��1���������Һ���ա�����������ȡ�½���ƿ����c2 mol��L��1NaOH����Һ�ζ���ʣ��HCl�����յ�ʱ����V2mLNaOH��Һ��
���ȵIJⶨ��ȷ��ȡ��ƷX�������Һ����AgNO3����Һ�ζ���K2CrO4��ҺΪָʾ���������ֵ���ɫ����������ʧΪ�յ㣨Ag2CrO4Ϊש��ɫ����
�ش��������⣺
��1��װ���а�ȫ�ܵ�����ԭ���� ��
��2����NaOH����Һ�ζ���ʣ��HClʱ��Ӧʹ�� ʽ�ζ��ܣ���ʹ�õ�ָʾ��Ϊ ��
��3����Ʒ�а���������������ʽΪ ��
��4���ⶨ��ǰӦ�ö�װ�ý��������Լ��飬�������Բ��òⶨ����� ���ƫ�ߡ���ƫ�͡�����
��5���ⶨ�ȵĹ����У�ʹ����ɫ�ζ��ܵ�ԭ���� ���ζ��յ�ʱ������Һ��c(Ag��)=2.0��10��5 mol��L��1��c(CrO42��)Ϊ mol��L��1������֪��Ksp(Ag2CrO4)=1.12��10��12��
��6�����ⶨ����ƷX���ܣ������ȵ����ʵ���֮��Ϊ1:6:3���ܵĻ��ϼ�Ϊ ���Ʊ�X�Ļ�ѧ����ʽΪ ��X���Ʊ��������¶Ȳ��ܹ��ߵ�ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��16�֣�ij��ѧ��ȤС�����ʵ�飬��Ũ������ͭ��Ӧ�Ƶ�SO2���������ʵ��̽����
��1��װ��A�з�����Ӧ�Ļ�ѧ����ʽ
��_______��װ��B��������_______��
���װ��C��Ŀ������֤SO2��
_______________�ԣ�D��NaOHȫ��ת��Ϊ
NaHSO3�ı�־��____________��
��3����Ӧ����ʱ��ƿ��Cu��ʣ�࣬ij
ͬѧ��ΪH2SO4Ҳ��ʣ�࣬�����������ʵ
�鷽�����ⶨʣ��H2SO4����������ȴ����
��ϡ�ͺ��������ʵ�飬�ܴﵽĿ����________������ţ���
a�� ������к͵ζ����ⶨ b�� ������Zn��Ӧ����������H2�����
c�� ��PH�Ʋ���ҺPHֵ d�� ������BaCl2��Һ��Ӧ���������ɳ���������
��4����Dƿ����NaHSO3��Һ�м���Ư����Һ����Ӧ�����ֿ��������
I�� HSO3-��ClO-�պ÷�Ӧ�ꣻ II�� Ư�۲��㣻 III�� Ư�۹���
ͬѧ�Ƿֱ�ȡ���������Һ���Թ��У�ͨ������ʵ��ȷ���÷�Ӧ������һ���������������±���
| ʵ����� | ʵ����� | ���� | ��Ӧ�Ŀ������ |
| �� | �μ��������۵⻯����Һ���� | | III |
| �� | �μ������غ�ɫ��KI3��Һ���� | | II |
| �� | �������������KMNO4��Һ���� | ��Һ����ɫ | |
| �� | ���뼸С��CaCO3���� | �����ݲ��� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijʵ��С�����÷�Ӧ2CuO��2Cl2 2CuCl2��O2�ⶨͭ�Ľ������ԭ���������ɹ�ѡ���װ����ͼ��ʾ��
2CuCl2��O2�ⶨͭ�Ľ������ԭ���������ɹ�ѡ���װ����ͼ��ʾ��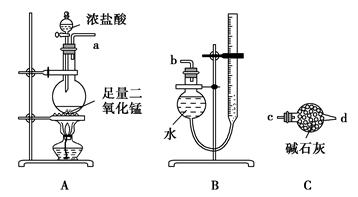

����һ��ͨ���ⶨ��Ӧ��CuO������m(CuO)�Ͳ���O2�����V(O2)���ⶨͭ�Ľ������ԭ��������
(1)����������������ý���(ͼ��δ����)��ѡ���������ϳ�һ��ʵ��װ�ã�����˳��Ϊa��(����)(����)��(����)(����)��(����)(����)��(����)(����)��b��
(2)װ��B���ɸ���ܺͼ�ʽ�ζ��ܸ�����ɵIJ������������װ�ã�ʵ��ǰ�ζ���Һ�������ΪV1 L��ʵ���ָ������£�����װ������Һ����ƽ��õ�ĩ����ΪV2 L��������ʱ����Ħ�����ΪVm L��mol��1����Eװ����CuO������Ϊm1 g����ַ�Ӧ������CuCl2������Ϊm2 g����ͭ�Ľ������ԭ�������ı���ʽΪ
[�ú�m1��V1��V2�Ĵ���ʽ��ʾ]��
(3)������ͭ�л���ͭ����ⶨ��� (�ƫ����ƫС������Ӱ�족)��
(4)װ��E��ʵ������е���Ҫ������ ��
������������A��D��E��F����װ��(β����������װ�ô���)��ɲⶨ����
(5)����Ϊ�ⶨ���������� (д��һ��)������ⶨ����������д��ͭ�Ľ������ԭ�������ı���ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ͼ��ʾ����ʵ���ҽ��а��������Ʊ�������ʵ������װ�ã����̶ֹ�װ��δ������
��1������װ��װ�ú���Ҫ����A��Eװ�õ������ԣ������������ ��Ȼ����A��???�쵽E��������ð�����ƿ��ƾ��ƻ��ɿ�˫�֣�E�е�����ˮ���γ�˵��װ�����������á�
��2��װ��B��ʢ���Լ��� ��
��3����ȼC���ƾ��ƣ��رյ��ɼ�2�����ɼ�1���ӷ�Һ©���ų�Ũ��ˮ����û��ƿ�й����رշ�Һ©�����Ժ�Ƭ�̣�װ��C�к�ɫ������죬װ��E����Һ����ִ������ݣ�ͬʱ���� ����������E���ݳ�Һ����������ֱ�������������д����C�з�����Ӧ�Ļ�ѧ����ʽ ��
��4����C�й���ȫ�����ɫ�رյ��ɼ�1�������ƿ��ƾ��ƣ�����ȴ����C�й�������������Ӧǰ��������Ϊ16g����Ӧ����ع�����������2.4g��ͨ������ȷ���ù������ijɷ��� ���û�ѧʽ��ʾ����
��5���ڹرյ��ɼ�1���ɼ�2�������������F�У��ܿ췢��װ��F�в������̣�ͬʱ����G����ҺѸ�ٵ�������F�С�д���������̵Ļ�ѧ����ʽ ��Ѹ�ٲ���������ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��15�֣�ij�о���ѧϰС���һ�������ǡ�NO2�ܷ�֧��ľ����ȼ�գ�������ʵ������û���ֳɵ�NO2���壬��С���ͬѧ�������������������N2O4�Ĵ��ڣ�ͼ������̨�ȼг�����������ȥ����
I��ʵ��װ������ͼ��ʾ��5�֣�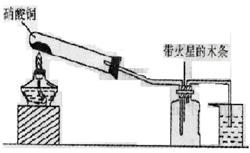
��1������ͭ������ȷֽ�IJ���������ͭ������������������д���÷�Ӧ�Ļ�ѧ����________________��
��2��������ƿ�г�������ɫ����ʱ��ľ����ȼ�ˡ��е�ͬѧ�ó���NO2��֧��ľ����ȼ�ա��Ľ��ۡ�����Ϊ��һ�����Ƿ���ȷ��____________�����ȷ������ȷ������������_______
II��ʵ��װ����ͼ��ʾ����5�֣�
��1��д��Ũ�������ȷֽ�Ļ�ѧ����ʽ��__________________________________����2�֣�
��2��ʵ�鿪ʼ��ľ���ϵĻ�����Ϩ���е�ͬѧ�����NO2����֧�� ľ����ȼ�ա��Ľ��ۡ�����Ϊ��һ�����Ƿ���ȷ��________�����ȷ������ȷ������������ ����3�֣�
III��Ϊ�˸���ֱ��˵����NO2�ܷ�֧��ľ����ȼ�ա���һ���⣬�����������һ����ʵ�鷽��������ʵ��ԭ������Ҫ��������2�֣�_________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
ij�о���ѧϰС��Ϊ��̽��þ������ˮ��Ӧ�Ļ�����������������ʵ�飺
�ٽ�þ��Ͷ����ˮ�У�δ���κ�����
�ڽ�þ�۷�����ˮ�У��۲쵽ֻ�ǿ�ʼʱ���������������ݣ�����ˮ����ɫ����ȥ��
�۽�þ�۷���Һ���У�δ�۲쵽�κ���������
��������þ�۵�Һ���еμӼ���ˮ���۲쵽��ĺ���ɫ�ܿ���ȥ��
�����й���þ����ˮ�ķ�Ӧ������������ȷ���ǣ� ��
| A��þ��ֱֻ������ˮ�е��巴Ӧ |
| B��þ��ֻ����ˮ�е��ᷴӦ |
| C������������������������þ����ˮ��Ӧ�õ� |
| D��þ����ˮ�Ĵ������巢����Ӧ |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com