
���ᣨH2C2O4)��������Ȼ���ֲ���У���K1=5.4��10-2����K2=5.4��10-5�����л�ԭ�ԣ�����ˮ����Һ�����ԣ�Ϊ�ⶨijH2C2O4��Һ��Ũ�ȣ�ȡ����Һ����ƿ�У���������ϡH2SO4����Ũ��Ϊc mol/L KMnO4����Һ�ζ����ζ�ԭ��Ϊ��2KMnO4+5H2C2O4+3H2SO4=K2SO4+10CO2��+2MnSO4+8H2O
��1���ζ�����ʹ��֮ǰ��������еIJ����� ���ζ�ʱ��KMnO4��ҺӦװ�� �����ʽ�ζ��ܡ���ʽ�ζ��ܡ����У��ﵽ�ζ��յ�ʱ������Ϊ ��
��2����ͼ��ʾ50mL�ζ�����Һ���λ�ã���A��C�̶ȼ����1mL��C���Ŀ̶�Ϊ20���ζ�����Һ�����ӦΪ mL����ʱ�ζ�����Һ������ 30.60 mL��(����ڡ�С�ڻ����)
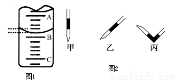
��3��Ϊ�˼�Сʵ������ͬѧһ������������ʵ�飬����ÿ����ȡH2C2O4��Һ�����ΪVmL������ʵ������¼���£�
ʵ����� | ��һ�� | �ڶ��� | ������ |
����KMnO4��Һ���/mL | 22.32 | 24.39 | 24.41 |
���ϱ����Կ�������һ��ʵ���м�¼����KMnO4��Һ������������ں����Σ���ԭ�������_
A��ʵ�����ʱ���ӿ̶��߶�ȡ�ζ��յ�ʱKMnO4��Һ�����
B���ζ�ǰ�ζ��ܼ��������ݣ��ζ�����������
C����һ�εζ�ʢװ��Һ�ĵζ���װҺǰ������ˮ��ϴ����δ�ñ�Һ��ϴ��
D����һ�εζ��õ���ƿ�ô�װҺ��ϴ����������δ��ϴ��
E���μ�KMnO4��Һ���죬δ������տ�����Һ��ɫ������ֹͣ�ζ�
��4�������������ݣ�д��H2C2O4�����ʵ���Ũ�ȵı���ʽ�����뻯��C=
��5������һ����Ƽ�ʵ��֤�����������ǿ��̼�ᣬʵ�������������

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2016��ɽ��ʡ������ѧ�����нβ��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
����ʵ�����������ͽ��۾���ȷ����
ѡ�� | ʵ����� | ���� | ���� |
A | SO2ͨ����ˮ�� | ��Һ��ɫ | SO2��Ư���� |
B | ��Fe(NO3)2��Ʒ����ϡH2SO4���μ�KSCN��Һ | ��Һ��� | Fe(NO3)2��Ʒ���������� |
C | ��̼������Һ��ͨ��CO2 | �о������� | �ܽ�ȣ� NaHCO3��Na2CO3 |
D | ��ʢ������NaHCO3���Թ��еμӲ�����Һ | �����ݲ��� | ���ԣ����̼�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ������ʡ��һ��ѧ�����в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
���з�Ӧ�У�����������ԭ��Ӧ����
��H2+Cl2 2HCl ��Na2CO3+2HCl�T2NaCl+H2O+CO2��
2HCl ��Na2CO3+2HCl�T2NaCl+H2O+CO2��
��2H2O 2H2��+O2����CuO+2HNO3�TCu��NO3��2+H2O
2H2��+O2����CuO+2HNO3�TCu��NO3��2+H2O
��2HgO 2Hg+O2��
2Hg+O2��
A���ڢ� B�� �٢� C�� �٢ڢ� D�� �ܢ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ��ɽ��ʡ�߶���ѧ�����в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
���ӣ�Ni-Cd���ɳ�������ִ��������й㷺Ӧ�á���֪ij���ӵ�صĵ������ҺΪKOH��Һ����䡢�ŵ簴��ʽ���У�
Cd + 2NiOOH + 2H2O
 Cd(OH��2 + 2Ni(OH��2�йظõ�ص�˵����ȷ����
Cd(OH��2 + 2Ni(OH��2�йظõ�ص�˵����ȷ����
A�����ʱ������Ӧ��Ni(OH��2��e- + OH-��NiOOH + H2O
B���������ǻ�ѧ��ת��Ϊ���ܵĹ���
C���ŵ�ʱ����������Һ�ļ��Բ���
D���ŵ�ʱ�������Һ�е�OH-�������ƶ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016�츣��ʡ�ĵ���У������ѧ�ڵڶ���������ѧ�Ծ��������棩 ���ͣ�ѡ����
����˵������ȷ����
A��PM 2.5��������̼�����ڿ��������ձ�������
B����ú�м�������CaSO4���ɴ�����ȼ�ղ�����SO2����
C���ԡ��ع��͡����з���ɵõ�����
D�����⻯ѧ�������������������ꡱ���γɶ��뵪���������й�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�����ʡ�߶���ѧ�����в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
���ܱ������е�һ����������巢����Ӧ��xA(g)+yB(g) zC(g),ƽ��ʱ���A��Ũ��Ϊ0.50mol/L�������¶Ȳ��䣬���������ݻ�����ԭ����2������ƽ��ʱ�����A��Ũ�Ƚ���Ϊ0.30mol/L�������й��ж���ȷ����
zC(g),ƽ��ʱ���A��Ũ��Ϊ0.50mol/L�������¶Ȳ��䣬���������ݻ�����ԭ����2������ƽ��ʱ�����A��Ũ�Ƚ���Ϊ0.30mol/L�������й��ж���ȷ����
A��x+y<z
B��ƽ��������Ӧ�����ƶ�
C��B��ת��������
D��C�������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�����ʡ�߶�������������ѧ�Ծ��������棩 ���ͣ�ѡ����
��֪�¶�Tʱˮ�����ӻ�����ΪKW�����¶��£���Ũ��Ϊa mol/L��һԪ��HA��b mol/L��һԪ��BOH�������ϣ����ж�����Һ�����Ե�������
A��a��b
B�������Һ��pH��7
C�������Һ�У�c(H��)��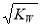 mol/L
mol/L
D�������Һ�У�c(H��) + c(B��) = c(OH��) + c(A��)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016�츣��ʡ������ѧ�ڵڶ����¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
��ͼ�����ڸ���ռ������ն��������װ�ã����з�����ȷ����
ѡ�� | X | �ռ����� | Y |
A | ��ʯ�� | �Ȼ��� | ˮ |
B | ��ʯ�� | ���� | ˮ |
C | �Ȼ��� | �������� | �������� |
D | �Ȼ��� | һ������ | �������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ��ɽ��ʡ�Ͳ��и�һ��ѧ�����в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
����ͬ�¶Ⱥ�ѹǿ�£�1molO2��1molO3��Ƚ�����˵���������
A�������ͬ������һ����22.4L
B����������ͬ����ԭ������ͬ
C��Ħ��������ͬ����������ͬ
D����������ͬ��������Ҳ��ͬ
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com