��2012?ɽ��ģ�⣩A��B��C����ǿ����ʣ�������ˮ�е�������������±���ʾ��
| ������ |
Na+��K+��Cu2+ |
| ������ |
SO42-��OH- |
��ͼ1��ʾװ���У��ס��ҡ��������ձ����ηֱ�ʢ��������A��Һ��������B��Һ��������C��Һ���缫��Ϊʯī�缫��

��ͨ��Դ������һ��ʱ��������c�缫����������16g�������¸��ձ�����Һ��pH����ʱ��t�Ĺ�ϵͼ��ͼ2���ݴ˻ش��������⣺
��1��MΪ��Դ��
��
��
�����������������
�缫b�Ϸ����ĵ缫��ӦΪ
4OH--4e-=2H2O+O2��
4OH--4e-=2H2O+O2��
��
��2������缫e�����ɵ������ڱ�״̬�µ������
5.6L
5.6L
��
��3��д�����ձ��ĵ��ط�Ӧ
2CuSO
4+2H
2O
2Cu+O
2��+2H
2SO
42CuSO
4+2H
2O
2Cu+O
2��+2H
2SO
4��4��������һ��ʱ��������c�缫����������16g��Ҫʹ���ָ���ԭ����״̬�������������
CuO
CuO
������
20g
20g
��
��5�������������B��Һ�еĽ�������ȫ������ʱ������Դ�缫���ӣ����һ��ʱ����ҳ�����Һ��pH
����
����
���������С�����䡱�����缫d�ϵķ�Ӧ��
Cu2++2e-=Cu
Cu2++2e-=Cu
��




 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
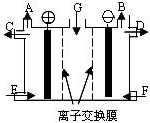 ��2012?ɽ��ģ�⣩���������ӽ���Ĥ��ֻ������Ӧ������ͨ������ʯī���缫�ĵ��۵�ⱥ�͵�Na2SO4��Һ����NaOH��H2SO4������˵������ȷ���ǣ�������
��2012?ɽ��ģ�⣩���������ӽ���Ĥ��ֻ������Ӧ������ͨ������ʯī���缫�ĵ��۵�ⱥ�͵�Na2SO4��Һ����NaOH��H2SO4������˵������ȷ���ǣ�������