
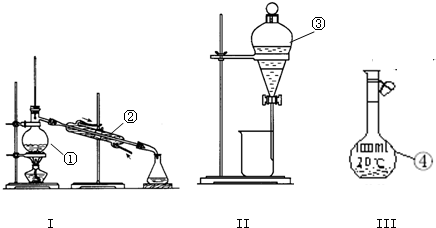
��18�֣���1��д����ͼ����Ţ١������������ƣ�
��2�������١����У�ʹ��ʱ�������Ƿ�©ˮ���� ������������ţ�
��3�������ˮ�еĵ�Ӧ��ѡ��װ�� ����װ����ţ����в���A����ѡװ�ý��� ���������в���Aʱ�����ڵ�ˮ�м�һ�Լ�����ѡ���Լ�ʱ��������������Ϊ��Щ�����DZ���ģ� (�����)��
�ٳ�����ΪҺ̬ ��I2�������ܽ�̶ȴ� ����ˮ���̶ܳ�С ���ܶ�Ҫ��ˮ��
��4����ͼ��ijѧУʵ���Ҵӻ�ѧ�Լ��̵���ص�Ũ�����Լ���ǩ�ϵIJ������ݡ����ø�Ũ��������480 mL 1 mol/L��ϡ���ᡣ
�ɹ�ѡ�õ������У�
�ٽ�ͷ�ιܣ�����ƿ�����ձ����� ҩ�ף�����Ͳ����������ƽ��
��ش��������⣺
a��ʢ��Ũ������Լ�ƿ��ǩ��Ӧӡ�����о�ʾ����е� 
b������ϡ����ʱ����ȱ�ٵ������� �� (д��������)��
c�������㣬����480 mL 1 mol/L��ϡ������Ҫ����Ͳ��ȡ����Ũ��������Ϊ______ mL����ȡ����ʱӦѡ�� mL������Ͳ��
A��10 mL B��50 mL C��100 mL D��200mL
d���������Ƶ�ϡ������вⶨ��������Ũ�ȴ���1 mol/L�����ƹ��������и�������������������ԭ���� ( ) ��
A������Ͳ��ȡŨ����ʱ�����ӿ̶���ȡŨ����
B������ʱ����������ƿ�̶��߽��ж���
C����ϡ�ͺ��ϡ��������ת������ƿ�����žͽ����Ժ��ʵ�����
D��ת����Һʱ��������������Һ��������ƿ����
E������ƿ������ˮϴ�Ӻ�δ���������������ˮ
F�����ݺ�����ƿ����ҡ�Ⱥ���Һ����ڿ̶��ߣ��㲹�伸��ˮ���̶ȴ�
 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��1��д����ͼ����Ţ١������������ƣ�
![]()
![]()

I II III
�� ���� �� �� ���� ��
��2�������١����У�ʹ��ʱ�������Ƿ�©ˮ���� ������������ţ�
��3�������ˮ�еĵ�Ӧ��ѡ��װ�� ����װ����ţ� ���� �� ���� ������װ�� ����װ����ţ����� ������
��4��������98%��Ũ����(�ܶ�Ϊ1.84g��cm��3)���Ƴ�Ũ��Ϊ0.5mol��L��1��ϡ����100mL��
�������������ձ������������ __________�� __________�� __________��
����ȡŨ��������Ϊ____________mL��
�����в�������������ҺŨ��ƫ�ߵ���
AȡŨ����ʱ����
B��Ũ���ᵹ����ϴ��װ�ã�����ϴ��Һ�����ձ���
C���ձ���ϡ��Ũ���������ת��
D����ʱ����
E �ߵ�ҡ�Ⱥ���Һ����ڿ̶��ߣ���δ��ˮ���̶���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015���㽭ʡ��һ10���¿���ѧ�Ծ��������棩 ���ͣ�ʵ����
��18�֣���1��д����ͼ����Ţ١������������ƣ�

��2�������١����У�ʹ��ʱ�������Ƿ�©ˮ���� ������������ţ�
��3�������ˮ�еĵ�Ӧ��ѡ��װ�� ����װ����ţ� ���в���A����ѡװ�ý��� ���������в���Aʱ�����ڵ�ˮ�м�һ�Լ�����ѡ���Լ�ʱ��������������Ϊ��Щ�����DZ���ģ� (�����)��
�ٳ�����ΪҺ̬ ��I2�������ܽ�̶ȴ� ����ˮ���̶ܳ�С ���ܶ�Ҫ��ˮ��
��4����ͼ��ijѧУʵ���Ҵӻ�ѧ�Լ��̵���ص�Ũ�����Լ���ǩ�ϵIJ������ݡ����ø�Ũ��������480 mL 1 mol/L��ϡ���ᡣ

�ɹ�ѡ�õ������У�
�ٽ�ͷ�ιܣ�����ƿ�����ձ����� ҩ�ף�����Ͳ����������ƽ��
��ش��������⣺
a��ʢ��Ũ������Լ�ƿ��ǩ��Ӧӡ�����о�ʾ����е�

b������ϡ����ʱ����ȱ�ٵ������� �� (д��������)��
c�������㣬����480 mL 1 mol/L��ϡ������Ҫ����Ͳ��ȡ����Ũ��������Ϊ______ mL����ȡ����ʱӦѡ�� mL������Ͳ��
A��10 mL B��50 mL C��100 mL D��200mL
d���������Ƶ�ϡ������вⶨ��������Ũ�ȴ���1 mol/L�����ƹ��������и�������������������ԭ���� ( ) ��
A������Ͳ��ȡŨ����ʱ�����ӿ̶���ȡŨ����
B������ʱ����������ƿ�̶��߽��ж���
C����ϡ�ͺ��ϡ��������ת������ƿ�����žͽ����Ժ��ʵ�����
D��ת����Һʱ��������������Һ��������ƿ����
E������ƿ������ˮϴ�Ӻ�δ���������������ˮ
F�����ݺ�����ƿ����ҡ�Ⱥ���Һ����ڿ̶��ߣ��㲹�伸��ˮ���̶ȴ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�������ʡ��һ10�·��¿���ѧ�Ծ� ���ͣ�ʵ����
��1��д����ͼ����Ţ١������������ƣ�
I II III
�� ���� �� �� ���� ��
��2�������١����У�ʹ��ʱ�������Ƿ�©ˮ���� ������������ţ�
��3�������ˮ�еĵ�Ӧ��ѡ��װ�� ����װ����ţ� ���� �� ���� ������װ�� ����װ����ţ����� ������
��4��������98%��Ũ����(�ܶ�Ϊ1.84g��cm��3)���Ƴ�Ũ��Ϊ0.5mol��L��1��ϡ����100mL��
�������������ձ������������ __________�� __________�� __________��
����ȡŨ��������Ϊ____________mL��
�����в�������������ҺŨ��ƫ�ߵ���
A ȡŨ����ʱ����
B ��Ũ���ᵹ����ϴ��װ�ã�����ϴ��Һ�����ձ���
C ���ձ���ϡ��Ũ���������ת��
D ����ʱ����
E �ߵ�ҡ�Ⱥ���Һ����ڿ̶��ߣ���δ��ˮ���̶���
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com