

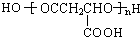 ��
��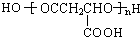 ��
��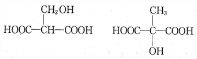 ��
��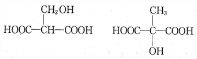 ��
��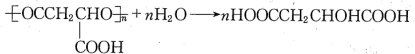 ��
��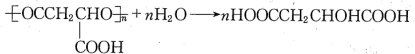 ��
��


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A��һ��D2O����������������Ϊ8 |
B��NH3�ĽṹʽΪ�� |
C��HCl�ĵ���ʽΪ |
| D�����ȶ��ԣ�HCl��HF |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 2003��10��16�ա�������š��ɴ��ɹ����䣬ʵ�����л�����ķ������룮���ͷɴ��Ļ��ȼ�ϳ�Һ̬˫��ˮ�⣬������һ��Һ̬���⻯�����֪�û���������Ԫ�ص���������Ϊ12.5%����Է�������Ϊ32���ṹ�������ָ÷��ӽṹ��ֻ�е�����
2003��10��16�ա�������š��ɴ��ɹ����䣬ʵ�����л�����ķ������룮���ͷɴ��Ļ��ȼ�ϳ�Һ̬˫��ˮ�⣬������һ��Һ̬���⻯�����֪�û���������Ԫ�ص���������Ϊ12.5%����Է�������Ϊ32���ṹ�������ָ÷��ӽṹ��ֻ�е������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ��� |
| �ŵ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A�����뱽������ɫ����Һ��K+��NH4+��Cl-��I-�������̪�ʺ�ɫ����Һ��SO42-��K+��Cl-��HSO3- �ֱ��ܴ������棨�����£� | ||
B��
| ||
| C����֪��25��ʱ��Mg��OH��2��kSP=5.61��10-12��MgF2��kSP=7.42��10-11��25��ʱ����Mg��OH��2������Һ�м���NaF��Һ��Mg��OH��2 ����ת��ΪMgF2 | ||
D����ʾH2ȼ���ȵĻ�ѧ����ʽΪ��H2��g��+
|
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com