
Ϊ�˺������û�ѧ�ܣ�ȷ����ȫ���������������Ҫ��ֿ��ǻ�ѧ��Ӧ���ʱ䣬����ȡ��Ӧ��ʩ����ѧ��Ӧ���ʱ�ͨ����ʵ����вⶨ��Ҳ�ɽ����������㡣
(1)ʵ���ã�1 g�״��������г��ȼ�����ɶ�����̼��Һ̬ˮ�ͷų�22.7 kJ����������д���״�ȼ�յ��Ȼ�ѧ����ʽ__________________________________
(2)��֪��ӦCH3��CH3(g)�D��CH2=CH2(g)��H2(g)���йػ�ѧ���ļ������¡�
| ��ѧ�� | C��H | C=C | C��C | H��H |
| ����/kJ��mol��1 | 414.4 | 615.3 | 347.4 | 435.3 |

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��14�֣�
��.�ȼҵ�г������ӽ���Ĥ������Ƽ��ͼ1��ʾ����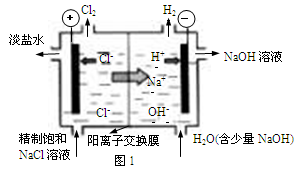
����1��д��ͼ1�������ĵ缫��Ӧʽ ��
����2����֪�����ӽ���Ĥֻ����������ͨ���������ӽ���Ĥֻ����������ͨ������ҵ������ͼ2װ�õ�ⱥ��Na2SO4��Һ������������NaOH��H2SO4�����װ������Ҫ��ȱ���� ��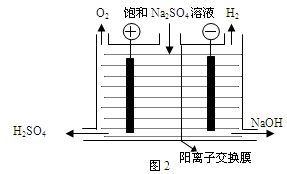
��.�����(MoS2)��һ����Ҫ�Ŀ��ͼ3�ǻ�����㱺��¯��ʾ��ͼ������1��2��3������¯���š�580��600��610�����Ǹ�¯����¶ȣ��棩��ͼ4�����˸�¯��������ϵ����ʵ����ٷֺ�����
��֪��MoS2��������1molMoO3�ķ�Ӧ�ȡ�H1=-1011KJ/mol��MoO2��������1molMoO3�ķ�Ӧ�ȡ�H2=-154KJ/mol���Իش�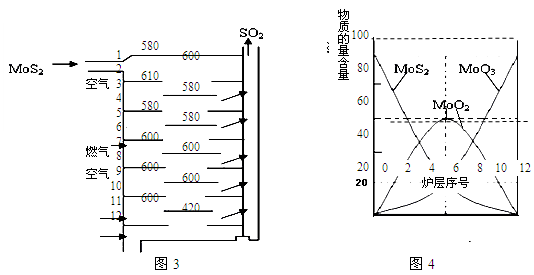
��1����֤����������ɵ�������SO2������SO3�ķ����� ��
��2������������ɵ�������ʹ�����ữ��KMnO4��Һ��ɫ���û�ѧ����ʽ��ʾ��ɫ��ԭ�� ��
��3����6¯����ڵĹ������ʷֱ���MoS2��MoO3��MoO2�������ǵ����ʵ���֮��Ϊ ��
��4��ͼ4�������м�¯�㣨4��6�����ܴ���һ�֡�����+���������+�����ķ�Ӧ����д���÷�Ӧ���Ȼ�ѧ��Ӧ����ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ĺϳ�������Ҫ�Ļ�������֮һ��
I����ҵ�Ϻϳɰ��õ�H2�ж�����ȡ�ķ�����
�� �ý�̿��ˮ��Ӧ�� C(s��+ H2O(g) CO(g��+ H2(g)��
CO(g��+ H2(g)��
�� ����Ȼ����ˮ������Ӧ��CH4(g��+ H2O(g�� CO (g)+ 3H2(g)
CO (g)+ 3H2(g)
��֪�йط�Ӧ�������仯����ͼ�������з�Ӧ�Ħ�H =__________ ___��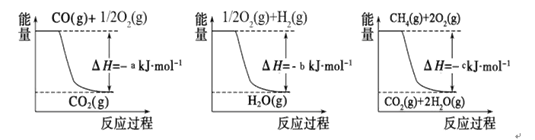
����3��1L���ܱ������У�ͬ�¶��¡�ʹ����ͬ�����ֱ���з�Ӧ��
3H2(g��+ N2(g�� 2NH3(g)������ͬ��ʽͶ�뷴Ӧ����ֺ��¡����ݣ���Ӧ�ﵽƽ��ʱ�й�����Ϊ��
2NH3(g)������ͬ��ʽͶ�뷴Ӧ����ֺ��¡����ݣ���Ӧ�ﵽƽ��ʱ�й�����Ϊ��
| �� �� | �� | �� | �� |
| ��Ӧ��Ͷ���� | 3 mol H2��2 mol N2 | 6 mol H2��4mol N2 | 2 mol NH3 |
| �ﵽƽ���ʱ�䣨min�� | t | 5 | 8 |
| ƽ��ʱN2��Ũ�ȣ�mol��L-1�� | c1 | 3 | |
| N2��������� | ��1 | ��2 | ��3 |
| ��������ܶȣ�g��L-1�� | ��1 | ��2 | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�о������ḻ��CO2��ȫ������Ϊ��̼Դ�������ǰӦ����㷺��̼Դ(ʯ�ͺ���Ȼ��)����������Ҷ���ݽߵ�Σ����ͬʱ�ֿɻ�����CO2�ۻ�������������ЧӦ��ʵ��CO2������ѭ����
��1��Ŀǰ��ҵ����һ�ַ�������CO2��H2��230�����������ת�����ɼ״�������ˮ��������ͼ��ʾ��ѹ������0.5 mol CO2��1.5 mol H2ת���ʴ�80%ʱ�������仯ʾ��ͼ��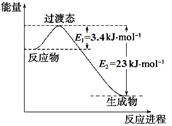
��д���÷�Ӧ���Ȼ�ѧ����ʽ�� ��
�����жϸ÷�Ӧ�ﵽ��ѧƽ��״̬�������� ��
a��������ѹǿ���� b��H2�������������
c��c(H2)=3c(CH3OH) d���������ܶȲ���
e��2��C��O���ѵ�ͬʱ��6��H��H���ѡ�
��2���˹���������ܹ�����̫���ܣ���CO2��H2O�Ʊ���ѧԭ�ϣ���ͼ��ͨ���˹���������Ʊ�HCOOHԭ����ʾ��ͼ������Ҫ��ش����⣺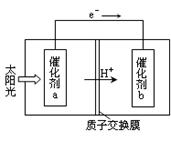
�ٸù����ǽ� ת��Ϊ ��(��������ѡ����ܡ���̫���ܡ�����ѧ�ܡ�)
�ڴ���b����ĵ缫��Ӧ����ʽΪ ��
��3��ij��������Ա�����ʹ�����������ͣ����ͻ�ѧʽ��C8H18��ʾ�����ȼ�ϵķ������Խ������CO2���ŷ����⡣�÷�����Ҫ���ô������CaH2����H2������������������������ʹ�ã�����ϵͳ�ֲ�������ȼ�ղ�����CO2����ϵͳ��Ӧ����ͼ��ʾ��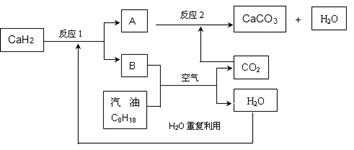
����������⣺
��д��CaH2�ĵ���ʽ ��
�ڷ�Ӧ1���������뻹ԭ�������ʵ���֮���ǣ� ��
�����ϵͳ��Ӧ��������ȫ����д����ϵͳ�ܷ�Ӧ�Ļ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������Ҫ�Ļ�����Ʒ�ͻ���ԭ�ϡ�
��1�����ĵ���ʽ�� ��
��2����֪��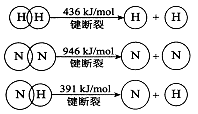
�ٺϳɰ����Ȼ�ѧ����ʽ�� ��
�ڽ����¶ȣ��÷�Ӧ�Ļ�ѧƽ�ⳣ��K �������������С�������䡱����
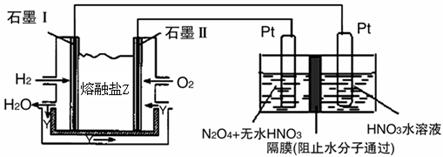
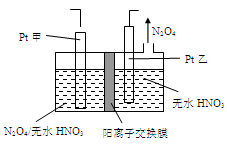
��3������������N2��H2Ϊ��Ӧ�������A��ϡ����Ϊ�������Һ��������������ṩ���ܣ����̵ܹ�������ȼ�ϵ�أ�װ����ͼl��ʾ��
��������ĵ缫��Ӧʽ�� ��A�� ��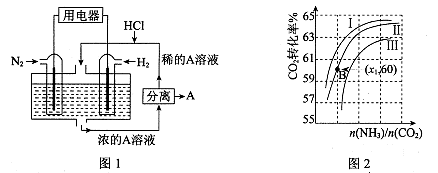
��4���ð��ϳ����صķ�ӦΪ2NH3(g)+CO2(g) CO(NH2)2��l��+ H2O(g)����ҵ����ʱ��ԭ��������ˮ������ͼ2��ʾCO2��ת�����백̼��
CO(NH2)2��l��+ H2O(g)����ҵ����ʱ��ԭ��������ˮ������ͼ2��ʾCO2��ת�����백̼��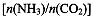 ��ˮ̼��
��ˮ̼��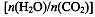 �ı仯��ϵ��
�ı仯��ϵ��
������I��II��III��Ӧ��ˮ̼�������� ��
�ڲ��B�㰱��ת����Ϊ40%����x1 ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���Ȼ���(TiCl4)����ȡ���캽�չ�ҵ���ϡ����ѺϽ����Ҫԭ�ϡ���������(��Ҫ�ɷ���FeTiO3)�Ʊ�TiCl4�Ȳ�Ʒ��һ�ֹ�������ʾ��ͼ���£�
(1)�����м�����м������Һ����ɫ����ʱ��Һ�Գ�ǿ���ԡ��ù����������·�Ӧ������
Fe��2Fe3��=3Fe2��
2TiO2��(��ɫ)��Fe��4H��=2Ti3��(��ɫ)��Fe2����2H2O
Ti3��(��ɫ)��Fe3����H2O=TiO2��(��ɫ)��Fe2����2H��
������������� ��
(2)�ڢڡ��۹��չ�������Ҫ�����������γ�TiO2��nH2O�ܽ������ܽ��ķ�ɢ�ʿ���ֱ����С�� ��Χ��
(3)���Ѣ����ƵõĹ���TiO2��nH2O������ϴ��ȥ���е����ʣ������Ƶ��Ѱۡ���֪25 ��ʱ��Ksp[Fe(OH)3]��2.79��10��39�����¶��·�ӦFe(OH)3��3H�� Fe3����3H2O��ƽ�ⳣ��K�� ��
Fe3����3H2O��ƽ�ⳣ��K�� ��
(4)��֪��TiO2(s)��2Cl2(g)=TiCl4(l)��O2(g) ��H����140 kJ��mol��1
2C(s)��O2(g)=2CO(g)����H����221 kJ��mol��1
д������TiO2�ͽ�̿��������Ӧ����Һ̬TiCl4��CO������Ȼ�ѧ����ʽ�� ��
(5)�������վ��гɱ��͡����õ�Ʒλ����Ϊԭ�ϵ��ŵ㡣������ɫ��ѧ����ù��������д��ڵIJ���֮���� (ֻҪ��д��һ��)��
(6)�����±���Ϣ��Ҫ���ƺ�����SiCl4���ʵ�TiCl4���ɲ��� ������
| | TiCl4 | SiCl4 |
| �۵�/�� | ��25.0 | ��68.6 |
| �е�/�� | 136.4 | 57.6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
��1����֪��N2(g)+O2(g) = 2NO(g) ��H=+180��5kJ/mol
N2(g)+3H2(g) 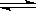 2NH3(g) ��H=��92��4kJ/mol
2NH3(g) ��H=��92��4kJ/mol
2H2(g)+O2(g) = 2H2O(g) ��H=��483��6kJ/mol
д����������������ȫ����һ�����������ˮ�������Ȼ�ѧ����ʽΪ
��
��2��ij����С���о����������������������£��ı���ʼ�����������ʵ�����N2(g)+3H2(g) 2NH3(g)��Ӧ��Ӱ�졣
2NH3(g)��Ӧ��Ӱ�졣
ʵ������ͼ��ʾ��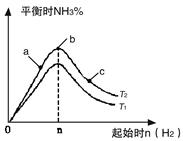
��ͼ��T��ʾ�¶ȣ�n��ʾ���ʵ�����
��ͼ����T2��T1�Ĺ�ϵ�ǣ�T2 T1������ڡ������ڡ������ڡ�����ȷ������
�ڱȽ���a��b��c����������ƽ��״̬�У���Ӧ��N2 ��ת������ߵ��ǣ�����ĸ���� ��
���������ݻ�Ϊ1L������ʼ��ϵ�м���1mol N2 ��n=3mol��Ӧ�ﵽƽ��ʱH2��ת����Ϊ60%����� �����£�T2������Ӧ��ƽ�ⳣ��K= ����������������䣬���������м���1mol N2��3mol H2��Ӧ�ﵽƽ��ʱ��������ת���ʽ�
����������������䡱����
��3��N2O5��һ�������������������ʺ��Ʊ��ܵ����ǵĹ�ע��
��һ���¶��£��ں����ܱ�������N2O5�ɷ������з�Ӧ��
2N2O5(g) 4NO2(g)��O2(g) ��H��0�±�Ϊ��Ӧ��T1�¶��µIJ���ʵ������
4NO2(g)��O2(g) ��H��0�±�Ϊ��Ӧ��T1�¶��µIJ���ʵ������
| t/s | 0 | 50 | 100 |
| c(N2O5)/mol��L��1 | 5��0 | 3��5 | 2��4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪��ѧ����������ʽ���ܿ����ת������д�±��Ŀհ�:
| ��ѧ��Ӧ����ʽ(����) | ����ת����ʽ |
| �� | �ɻ�ѧ��ת��Ϊ���� |
��Pb+PbO2+2H2SO4 2PbSO4+2H2O 2PbSO4+2H2O | |
��CaCO3 CaO+CO2�� CaO+CO2�� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�п�Ժ�����о����о�Ա���ʽ���������ͬ�к������Ա�������PM2.5��ѧ��ɼ���Դ�ļ��ڱ仯�о����֣�����PM2.5��6����Ҫ��Դ�����У�����β����ȼú�ֱ�ռ4%��18%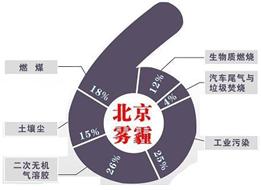
��1�����ھ�������β���ķ�ӦΪ��2NO(g)+2CO(g)
 2CO2(g)+N2(g)����֪�÷�Ӧ��570Kʱ��ƽ�ⳣ��Ϊ1��1059������Ӧ���ʼ���������˵����ȷ���ǣ�________
2CO2(g)+N2(g)����֪�÷�Ӧ��570Kʱ��ƽ�ⳣ��Ϊ1��1059������Ӧ���ʼ���������˵����ȷ���ǣ�________
| A��װ��β������װ�õ������ų��������в��ٺ���NO��CO |
| B�����β������Ч�ʵij��÷����������¶� |
| C������ѹǿ������ƽ�����ƣ���ʵ�ʲ����п�ͨ����ѹ�ķ�ʽ����侻��Ч�� |
| D�����β������Ч�ʵ����;����ʹ�ø�Ч���� |

 Ni(CO)4(g)������CO��Ӧ������������ж���Ϊ��ֹ�������ж�����ҵ�ϳ���SO2��ȥCO��������ΪS��CO2����֪��ط�Ӧ���̵������仯��ͼ��ʾ
Ni(CO)4(g)������CO��Ӧ������������ж���Ϊ��ֹ�������ж�����ҵ�ϳ���SO2��ȥCO��������ΪS��CO2����֪��ط�Ӧ���̵������仯��ͼ��ʾ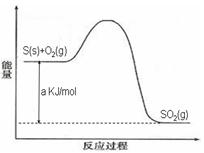
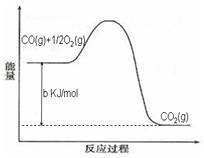
 2N2(g)+3H2O(g)��H��0��Ϊ��ߵ��������ת���ʿɲ�ȡ�Ĵ�ʩ�ǣ�������һ�֣�____________________��
2N2(g)+3H2O(g)��H��0��Ϊ��ߵ��������ת���ʿɲ�ȡ�Ĵ�ʩ�ǣ�������һ�֣�____________________��
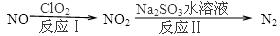
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com