
(6��)���������������������γ����������꣬�������γɹ⻯ѧ��������˱���Ժ��е�������ķ������д�����
(1)������������Һ�������շ����еĵ��������Ӧ�Ļ�ѧ����ʽ���£�
![]() ======
======![]()
�ڸ÷�Ӧ�У��������� ����ԭ���� ��
(2)����β���к���һ��������һ����̼�������������еĴ�ת����ת��Ϊ�Դ�������Ⱦ�����ʡ�д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
(3)��һ�������°��������������������ת��Ϊ����Ⱦ�����ʡ�д�������Ͷ���������һ�������·�Ӧ�Ļ�ѧ����ʽ�� ��
 ���Ǽ���С����ϵ�д�
���Ǽ���С����ϵ�д� �Ͻ�ƽ���Ȿϵ�д�
�Ͻ�ƽ���Ȿϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| c(H2)c(CO) |
| c(H2O) |
| �¶�/�� | 400 | 500 | 800 |
| ƽ�ⳣ��K | 9.94 | 9 | 1 |
 ��
�� ��ʵ����N-N������Ϊ167kJ?mol-1��NO2�е������ļ���Ϊ466kJ?mol-1��N2O4�е������ļ���Ϊ438.5kJ?mol-1����д��NO2ת��ΪN2O4���Ȼ�ѧ����ʽ
��ʵ����N-N������Ϊ167kJ?mol-1��NO2�е������ļ���Ϊ466kJ?mol-1��N2O4�е������ļ���Ϊ438.5kJ?mol-1����д��NO2ת��ΪN2O4���Ȼ�ѧ����ʽ�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����

| C(H2)C(CO) |
| C(H2O) |
| �¶�/�� | 400 | 500 | 800 |
| ƽ�ⳣ��K | 9.94 | 9 | 1 |
 ����֪N-N������Ϊ167kJ?mol-1��NO2�е������ļ���Ϊ466kJ?mol-1��N2O4�е������ļ���Ϊ438.5kJ?mol-1����д��NO2ת��ΪN2O4���Ȼ�ѧ����ʽΪ
����֪N-N������Ϊ167kJ?mol-1��NO2�е������ļ���Ϊ466kJ?mol-1��N2O4�е������ļ���Ϊ438.5kJ?mol-1����д��NO2ת��ΪN2O4���Ȼ�ѧ����ʽΪ�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
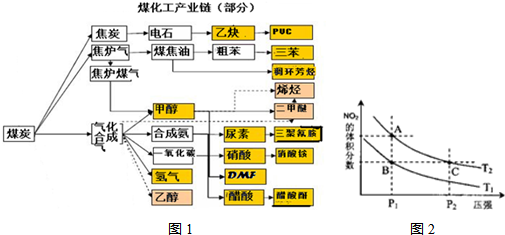
��16�֣���ͼ��ú������ҵ����һ���֣���������ѧ֪ʶ������������⣺
��1���ò�ҵ���кϳɰ��ķ�Ӧ�ڽϵ������ܷ��Է����У� ��
��2����֪�ò�ҵ����ij��Ӧ��ƽ�����ʽΪ��
������Ӧ�Ļ�ѧ��ӦΪ�� ��
��3����֪��һ���¶��£�����Ӧ��ƽ�ⳣ�����£�
C��s��+CO2��g�� 2CO��g����K1
CO��g��+H2O��g H2��g��+CO2��g���� K2
C��s��+H2O��g�� CO��g��+H2��g�� ��K3
��K1��K2��K3֮��Ĺ�ϵ�ǣ� ��
��4��ú����ͨ��ͨ���о���ͬ�¶���ƽ�ⳣ���Խ������ʵ�����⡣��֪�������һ����̼��ˮ�������뷴Ӧ��ʱ���ᷢ�����·�Ӧ��CO(g)+H2O(g) H2(g)+CO2(g)���÷�Ӧƽ�ⳣ�����¶ȵı仯���£�
| �¶�/�� | 400 | 500 | 800 |
| ƽ�ⳣ��K | 9.94 | 9 | 1 |
�÷�Ӧ������Ӧ������ ��Ӧ������ȡ����ȡ���������500��ʱ���У�����ʼʱCO��H2O����ʼŨ�Ⱦ�Ϊ0.020mol/L���ڸ������£�CO��ƽ��ת����Ϊ�� ��
��5����ͼ�п��������������������ᣬ�˹������漰���������NO��NO2��N2O4�ȡ���֪NO2��N2O4�Ľṹʽ�ֱ�����
����֪N��N������Ϊ167kJ��mol��1��NO2�е������ļ���Ϊ466kJ��mol��1��N2O4�е������ļ���Ϊ438.5kJ��mol��1����д��NO2ת��ΪN2O4���Ȼ�ѧ����ʽΪ ��
�Է�ӦN2O4(g)2NO2(g)�����¶�ΪT1��T2ʱ��ƽ����ϵ��NO2�����������ѹǿ�仯������ͼ��ʾ��
����˵����ȷ���ǡ���������
A��A��C����ķ�Ӧ���ʣ�A��C
B��A��C�����������ɫ��A�Cdz
C��B��C����������ƽ����Է���������B��C
D����״̬B��״̬A�������ü��ȵķ���
��6����������ҵ���м״�Ϊȼ���Ƴ�ȼ�ϵ�أ���д�����������ؽ����иõ�صĸ�����Ӧʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(6��)���������������������γ����������꣬�������γɹ⻯ѧ��������˱���Ժ��е�������ķ������д�����
(1)������������Һ�������շ����еĵ��������Ӧ�Ļ�ѧ����ʽ����:
![]() ======
======![]()
�ڸ÷�Ӧ�У��������� ����ԭ���� ��
(2)����β���к���һ��������һ����̼�������������еĴ�ת����ת��Ϊ�Դ�������Ⱦ�����ʡ�д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
(3)��һ�������°��������������������ת��Ϊ����Ⱦ�����ʡ�д�������Ͷ���������һ�������·�Ӧ�Ļ�ѧ����ʽ�� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com