
 –¥≥ωΡΨΧΩ”κ≈®ΝρΥαΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ
–¥≥ωΡΨΧΩ”κ≈®ΝρΥαΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ
| ||
| ||
| ||
| ||


 Οϊ–ΘΩΈΧΟœΒΝ–¥πΑΗ
Οϊ–ΘΩΈΧΟœΒΝ–¥πΑΗ
| ΡξΦΕ | ΗΏ÷–ΩΈ≥Χ | ΡξΦΕ | ≥θ÷–ΩΈ≥Χ |
| ΗΏ“Μ | ΗΏ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ“Μ | ≥θ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏΕΰ | ΗΏΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θΕΰ | ≥θΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏ»ΐ | ΗΏ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ»ΐ | ≥θ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚ
Θ®11Ζ÷Θ©
ΔώΘ°ΦΉΆ§―ßάϊ”Οœ¬Ν–ΉΑ÷Ο―ι÷ΛΡΨΧΩ”κ≈®ΝρΥαΖ¥”ΠΒΡ»Ϊ≤Ω≤ζΈο
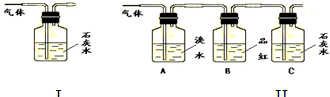
Θ®1Θ©–¥≥ωΡΨΧΩ”κ≈®ΝρΥαΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ
Θ®2Θ©A÷–Φ”»κΒΡ ‘ΦΝ « Θ§BΓΔD÷–Φ”»κΒΡ ‘ΦΝΕΦ «ΤΖΚλ»ή“ΚΘ§D÷–’ΐ»ΖΒΡ Β―ιœ÷œσ «ΘΚ
Θ®3Θ© Β―ι ±Θ§C÷–»τΦ”»κΒΡ «ΒΈ”–ΒμΖέΒΡΒβΥ°Θ§Ιέ≤λΒΫΒΡœ÷œσ « Θ§
άκΉ”ΖΫ≥Χ ΫΈΣΘΚ ΓΘ
ΔρΘ°““Ά§―ß÷Μ”ΟBΓΔCΓΔDΓΔEΉΑ÷Ο―ι÷ΛSO2ΒΡΡ≥–©–‘÷ Θ§«κΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®1Θ©C÷–Φ”»κΒΡ ‘ΦΝ « Θ§÷ΛΟςSO2ΨΏ”–―θΜ·–‘ΓΘ
Θ®2Θ©D÷–Φ”»κΥα–‘ΒΡKMnO4»ή“ΚΘ§÷ΛΟςSO2ΨΏ”– –‘ΓΘ
Θ®3Θ©E÷–Φ”»κΒΈ”–Ζ”ΧΣΒΡNaOH»ή“ΚΘ§÷ΛΟςSO2 « –‘ΤχΧεΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2010ΡξΥΡ¥® ΓΟΦ…Ϋ÷–―ßΗΏ“Μœ¬―ßΤΎΤΎΡ©ΫΧ―ß÷ ΝΩΦλ≤βΜ·―ß ‘Χβ Χβ–ΆΘΚ Β―ιΧβ
Θ®11Ζ÷Θ©
ΔώΘ°ΦΉΆ§―ßάϊ”Οœ¬Ν–ΉΑ÷Ο―ι÷ΛΡΨΧΩ”κ≈®ΝρΥαΖ¥”ΠΒΡ»Ϊ≤Ω≤ζΈο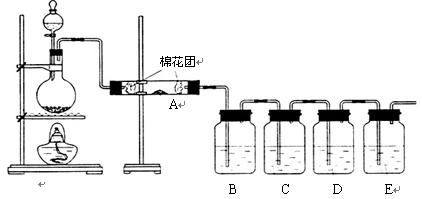
Θ®1Θ©–¥≥ωΡΨΧΩ”κ≈®ΝρΥαΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ
Θ®2Θ©A÷–Φ”»κΒΡ ‘ΦΝ « Θ§BΓΔD÷–Φ”»κΒΡ ‘ΦΝΕΦ «ΤΖΚλ»ή“ΚΘ§D÷–’ΐ»ΖΒΡ Β―ιœ÷œσ «ΘΚ
Θ®3Θ© Β―ι ±Θ§C÷–»τΦ”»κΒΡ «ΒΈ”–ΒμΖέΒΡΒβΥ°Θ§Ιέ≤λΒΫΒΡœ÷œσ « Θ§
άκΉ”ΖΫ≥Χ ΫΈΣΘΚ ΓΘ
ΔρΘ°““Ά§―ß÷Μ”ΟBΓΔCΓΔDΓΔEΉΑ÷Ο―ι÷ΛSO2ΒΡΡ≥–©–‘÷ Θ§«κΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®1Θ©C÷–Φ”»κΒΡ ‘ΦΝ « Θ§÷ΛΟςSO2ΨΏ”–―θΜ·–‘ΓΘ
Θ®2Θ©D÷–Φ”»κΥα–‘ΒΡKMnO4»ή“ΚΘ§÷ΛΟςSO2ΨΏ”– –‘ΓΘ
Θ®3Θ©E÷–Φ”»κΒΈ”–Ζ”ΧΣΒΡNaOH»ή“ΚΘ§÷ΛΟςSO2 « –‘ΤχΧεΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2010ΡξΥΡ¥® ΓΗΏ“Μœ¬―ßΤΎΤΎΡ©ΫΧ―ß÷ ΝΩΦλ≤βΜ·―ß ‘Χβ Χβ–ΆΘΚ Β―ιΧβ
Θ®11Ζ÷Θ©
ΔώΘ°ΦΉΆ§―ßάϊ”Οœ¬Ν–ΉΑ÷Ο―ι÷ΛΡΨΧΩ”κ≈®ΝρΥαΖ¥”ΠΒΡ»Ϊ≤Ω≤ζΈο
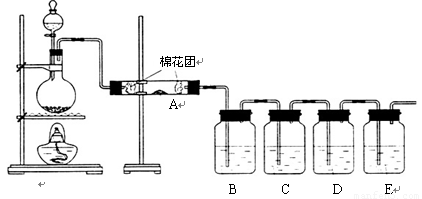
Θ®1Θ©–¥≥ωΡΨΧΩ”κ≈®ΝρΥαΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ
Θ®2Θ©A÷–Φ”»κΒΡ ‘ΦΝ « Θ§BΓΔD÷–Φ”»κΒΡ ‘ΦΝΕΦ «ΤΖΚλ»ή“ΚΘ§D÷–’ΐ»ΖΒΡ Β―ιœ÷œσ «ΘΚ
Θ®3Θ© Β―ι ±Θ§C÷–»τΦ”»κΒΡ «ΒΈ”–ΒμΖέΒΡΒβΥ°Θ§Ιέ≤λΒΫΒΡœ÷œσ « Θ§
άκΉ”ΖΫ≥Χ ΫΈΣΘΚ ΓΘ
ΔρΘ°““Ά§―ß÷Μ”ΟBΓΔCΓΔDΓΔEΉΑ÷Ο―ι÷ΛSO2ΒΡΡ≥–©–‘÷ Θ§«κΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®1Θ©C÷–Φ”»κΒΡ ‘ΦΝ « Θ§÷ΛΟςSO2ΨΏ”–―θΜ·–‘ΓΘ
Θ®2Θ©D÷–Φ”»κΥα–‘ΒΡKMnO4»ή“ΚΘ§÷ΛΟςSO2ΨΏ”– –‘ΓΘ
Θ®3Θ©E÷–Φ”»κΒΈ”–Ζ”ΧΣΒΡNaOH»ή“ΚΘ§÷ΛΟςSO2 « –‘ΤχΧεΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΫβ¥πΧβ
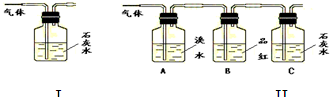 –¥≥ωΡΨΧΩ”κ≈®ΝρΥαΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ______Θ°
–¥≥ωΡΨΧΩ”κ≈®ΝρΥαΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ______Θ°≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΙζΦ ―ß–Θ”≈―Γ - ΝΖœΑ≤αΝ–±μ - ‘ΧβΝ–±μ
Κΰ±± ΓΜΞΝΣΆχΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΤΫΧ® | Άχ…œ”–ΚΠ–≈œΔΨΌ±®Ή®«χ | Βγ–≈’©Τ≠ΨΌ±®Ή®«χ | …φάζ Ζ–ιΈό÷ς“ε”–ΚΠ–≈œΔΨΌ±®Ή®«χ | …φΤσ«÷»®ΨΌ±®Ή®«χ
ΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΒγΜΑΘΚ027-86699610 ΨΌ±®” œδΘΚ58377363@163.com