

 ÿ��2�֣�
ÿ��2�֣� Al(OH)3(����)+3H+��
Al(OH)3(����)+3H+��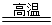 Fe3O4+4H2 �Ȼ�ԭ���������Ȼ�ԭ����
Fe3O4+4H2 �Ȼ�ԭ���������Ȼ�ԭ����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
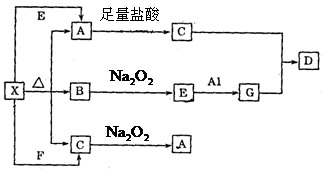
 ��G�������C��Ӧ����D�����ӷ���ʽ�� ��
��G�������C��Ӧ����D�����ӷ���ʽ�� �� A�����ӷ���ʽ�� ��
A�����ӷ���ʽ�� ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

 �Ļ�ѧʽ��A ��B ��C ��
�Ļ�ѧʽ��A ��B ��C ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��19���ͳ���Ӣ����ѧ�ҵ��������ԭ��ѧ˵ |
| B��1911�꣬¬ɪ�����ݦ�����ɢ����������˴��˵�ԭ�ӽṹģ�� |
| C����ķ������ԭ���д��ڵ��ӣ������ԭ�ӽṹ���ģ�� |
| D��20���ͳ�����ѧ��ָ��������������ѧ��������������ӵ��˶� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

 ��
���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

| | �ŵ� | ȱ�� |
| ʹ��Ũ���� | ��Ӧ���ʿ� | ��Ľϴ���NOx�����϶� |
| ʹ��ϡ���� | | |
 ��
�� ��
�� Ӧ�Ļ�ѧ����ʽΪ�� ��
Ӧ�Ļ�ѧ����ʽΪ�� ���鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com