
 ��֪NO2��N2O4�����ת����2NO2��g��?N2O4��g��������ӦΪ���ȷ�Ӧ�����ֽ�һ����NO2 ��N2O4 �Ļ������ͨ��һ���Ϊ1L�ĺ����ܱ������У���Ӧ���Ũ����ʱ��仯�Ĺ�ϵ������ͼ��ʾ����ش��������⣺
��֪NO2��N2O4�����ת����2NO2��g��?N2O4��g��������ӦΪ���ȷ�Ӧ�����ֽ�һ����NO2 ��N2O4 �Ļ������ͨ��һ���Ϊ1L�ĺ����ܱ������У���Ӧ���Ũ����ʱ��仯�Ĺ�ϵ������ͼ��ʾ����ش��������⣺ N2O4��g����֪��NO2��Ũ�ȱ仯��N2O4Ũ�ȱ仯����2�����ݴ˽��ͼ��������Ũ�ȵı仯���жϣ�
N2O4��g����֪��NO2��Ũ�ȱ仯��N2O4Ũ�ȱ仯����2�����ݴ˽��ͼ��������Ũ�ȵı仯���жϣ�| ��c |
| ��t |
| 0.4mol/L |
| 10min |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��֪NO2��N2O4�����ת����2NO2��g��?N2O4��g����H��0���ֽ�һ����NO2��N2O4�Ļ������ͨ��һ�����Ϊ2L�ĺ����ܱղ��������У���Ӧ��Ũ����ʱ��仯��ϵ��ͼ��ʾ��
��֪NO2��N2O4�����ת����2NO2��g��?N2O4��g����H��0���ֽ�һ����NO2��N2O4�Ļ������ͨ��һ�����Ϊ2L�ĺ����ܱղ��������У���Ӧ��Ũ����ʱ��仯��ϵ��ͼ��ʾ��| 10 |
| 9 |
| 10 |
| 9 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| c(H2)c(CO) |
| c(H2O) |
| �¶�/�� | 400 | 500 | 800 |
| ƽ�ⳣ��K | 9.94 | 9 | 1 |
 ��
�� ��ʵ����N-N������Ϊ167kJ?mol-1��NO2�е������ļ���Ϊ466kJ?mol-1��N2O4�е������ļ���Ϊ438.5kJ?mol-1����д��NO2ת��ΪN2O4���Ȼ�ѧ����ʽ
��ʵ����N-N������Ϊ167kJ?mol-1��NO2�е������ļ���Ϊ466kJ?mol-1��N2O4�е������ļ���Ϊ438.5kJ?mol-1����д��NO2ת��ΪN2O4���Ȼ�ѧ����ʽ�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��2009?��Ǩ��ģ����֪NO2��N2O4�����ת����2NO2��g��?N2O4��g����H��0���ں��������½�һ����NO2��N2O4�Ļ������ͨ��һ�ݻ�Ϊ2L���ܱ������У���Ӧ��Ũ����ʱ��仯��ϵ��ͼ������˵����ȷ���ǣ�������
��2009?��Ǩ��ģ����֪NO2��N2O4�����ת����2NO2��g��?N2O4��g����H��0���ں��������½�һ����NO2��N2O4�Ļ������ͨ��һ�ݻ�Ϊ2L���ܱ������У���Ӧ��Ũ����ʱ��仯��ϵ��ͼ������˵����ȷ���ǣ��������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
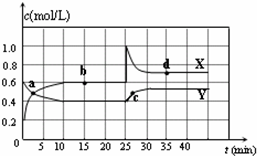 ��֪NO2��N2O4�����ת����2NO2��g��?N2O4��g����H��0���ں��������½�һ����NO2��N2O4�Ļ������ͨ��һ�ݻ�Ϊ2L���ܱ������У���Ӧ��Ũ����ʱ��仯��ϵ��ͼ������˵����ȷ���ǣ�������
��֪NO2��N2O4�����ת����2NO2��g��?N2O4��g����H��0���ں��������½�һ����NO2��N2O4�Ļ������ͨ��һ�ݻ�Ϊ2L���ܱ������У���Ӧ��Ũ����ʱ��仯��ϵ��ͼ������˵����ȷ���ǣ�������| A��ͼ�е��������ߣ�X�DZ�ʾNO2Ũ����ʱ��ı仯���� | B��a��b��c��d�ĸ����У�ֻ��b��d��Ļ�ѧ��Ӧ����ƽ��״̬ | C��25 minʱ������ƽ���ƶ���ԭ���ǽ��ܱ������������СΪ1L | D��ǰ10 min����v��NO2����ʾ�Ļ�ѧ��Ӧ����Ϊ0.06 mol/��L?min�� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com