
(15��)����ͼװ�ý���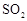 ת��Ϊ
ת��Ϊ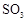 ��ת���ʲⶨʵ�飺
��ת���ʲⶨʵ�飺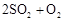
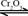
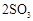 ��
��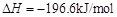 ����֪��
����֪��
| | �۵㣨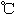 �� �� | �е㣨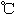 �� �� |
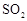 | -72.4 | -10 |
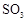 | 16.8 | 44.3 |

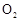 ��Ϊʹ
��Ϊʹ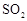 �нϸߵ�ת���ʣ�ʵ��ʱ����Ũ��������ȴ������Ⱥ�˳���� ��
�нϸߵ�ת���ʣ�ʵ��ʱ����Ũ��������ȴ������Ⱥ�˳���� ��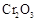 �������ʱ��Ӧ���ƾ����ƿ�һ����ټ��ȡ����Է��¶ȹ��ߣ���������ԭ���� �����ô�������
�������ʱ��Ӧ���ƾ����ƿ�һ����ټ��ȡ����Է��¶ȹ��ߣ���������ԭ���� �����ô�������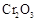 ���ķ�Ӧ��ʱ��
���ķ�Ӧ��ʱ��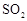 ��ת���ʻ� ������ߡ��������͡����䡱����
��ת���ʻ� ������ߡ��������͡����䡱����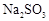 �������Ũ�����������ģ���Ӧ����ʱ����ͨ��
�������Ũ�����������ģ���Ӧ����ʱ����ͨ�� һ��ʱ�䣬�Ƶâ�װ�õ���������11.3g����ʵ����
һ��ʱ�䣬�Ƶâ�װ�õ���������11.3g����ʵ���� ��ת����Ϊ %������С�����һλ����
��ת����Ϊ %������С�����һλ����  ѧ�����νӽ̲��Ͼ���ѧ������ϵ�д�
ѧ�����νӽ̲��Ͼ���ѧ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
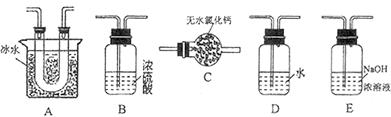
(15��)������ͼ��ʾװ�ü�����ҩƷ̽����ҵ������ʱ���������ת���ʡ���֪C�к�����n mol�����������C��Dʱ�ֱܷ���ȫ����ijһ�����壬�Һ���װ���ڿ����е�CO2��
��ش��������⣺
��1�������װ�õ��������Ҽ���ҩƷ��ʼ����ʱ����Ӧ���еIJ�����
��
��2��ͨ���۲������ݵ��ݳ����ʿɿ���SO2��O2������������ȣ�Aװ�û���������� ��
��3��Ϊ����߲ⶨSO2ת���ʵ�ȷ�ȣ����в�����Ľ����е��ǣ�����ţ� ��
��Ϩ��ƾ��ƺ�ֹͣͨ��SO2������ͨ��O2һ��ʱ��
��Ϩ��ƾ��ƺ�ֹͣͨ��O2������ͨ��SO2һ��ʱ��
����C��D֮���װһ��ʢ��Ũ�����ϴ��ƿ
��4�������徭����֣�ʵ�������ϡ��װ��C�е���Һ���������м���������BaCl2��Һ���õ��ij�������Ϊw g����װ��D���ӵ�����Ϊa g������������ת�����ǣ�
���ú���ĸ�Ĵ���ʽ��ʾ������ɲ�����
��5������Ӵ�����ͨ��2 molSO2(g)��1molO2(g)����һ���¶��£���Ӧ�ﵽƽ��ʱ��ýӴ����ڵ�ѹǿΪ��ʼʱ��0.75������SO2��ת����Ϊ ����ͬ�����£�����ʼʱͬʱͨ��a mol SO2��bmol O2��c mol SO3(g)ʱ���ﵽƽ��ʱ��ԭƽ���Ч������ʼʱά�ֻ�ѧ��Ӧ���淴Ӧ������У���c��ȡֵ��ΧΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ���㽭ʡ����ѧ�߶���ѧ����ĩ��ѧ�Ծ����������� ���ͣ�ʵ����
������þ(MgO2)������ϡ�ᣬ�����������������⣬��ҽѧ�Ͽ���Ϊ������ȡ�������þ��Ʒ�г����������MgO��ʵ���ҿ�ͨ�����ַ����ⶨ��Ʒ�й�����þ�ĺ�����
(1)ij�о�С��������ͼװ�òⶨһ����������Ʒ�й�����þ�ĺ�����
��ʵ��ǰ����еIJ����� ���� ��ϡ�����м�������FeCl3��Һ�������� �������� ��
���ú�ѹ��Һ©�����ŵ��У�ʹ��Һ©���е���Һ˳�����£� ��
��ʵ������ʱ�����ָ������£��� ���������� ����ƽ�ӿ̶��߶�����
(2)ʵ���һ���ͨ���������ַ����ⶨ��Ʒ�й�����þ�ĺ�����
����I��ȡa g��Ʒ����������ϡ���ᣬ��ַ�Ӧ���ټ��� NaOH��Һ��Mg2��������ȫ�����ˡ�ϴ�Ӻ�����������գ����յõ�b g���塣
��֪������Ksp[Mg(OH)2]=l��10��11��Ϊʹ����I��Mg2+��ȫ����[����Һ��c(Mg2+)��l ��10��5mol/L]����Һ��pH����Ӧ���� ��
�������õ�����ƽ��ȡ0.1000g��Ʒ������ƿ�У�����15 mL0.6 mol/LKI��Һ���������ᣬҡ�Ⱥ��ڰ�������5 min���������� ��ָʾ����Ȼ���� ��ʽ�ζ���ʢװ0. 1000 mol/L Na2S2O3��Һ���еζ�. (��֪��I2+2Na2S2O3= Na2S4O6+2NaI)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010������������ѧ������ѧ�ڵ��ߴ��¿������ۣ���ѧ���� ���ͣ�ʵ����
(15��)������ͼ��ʾװ�ü�����ҩƷ̽����ҵ������ʱ���������ת���ʡ���֪C�к�����nmol�����������C��Dʱ�ֱܷ���ȫ����ijһ�����壬�Һ���װ���ڿ����е�CO2��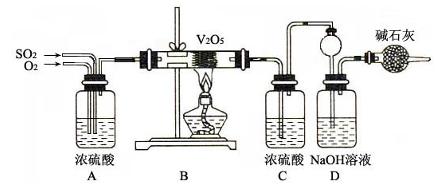
��ش��������⣺
��1�������װ�õ��������Ҽ���ҩƷ��ʼ����ʱ����Ӧ���еIJ�����
��
��2��ͨ���۲������ݵ��ݳ����ʿɿ���SO2��O2������������ȣ�Aװ�û���������� ��
��3��Ϊ����߲ⶨSO2ת���ʵ�ȷ�ȣ����в�����Ľ����е��ǣ�����ţ� ��
��Ϩ��ƾ��ƺ�ֹͣͨ��SO2������ͨ��O2һ��ʱ��
��Ϩ��ƾ��ƺ�ֹͣͨ��O2������ͨ��SO2һ��ʱ��
����C��D֮���װһ��ʢ��Ũ�����ϴ��ƿ
��4�������徭����֣�ʵ�������ϡ��װ��C�е���Һ���������м���������BaCl2��Һ���õ��ij�������Ϊw g����װ��D���ӵ�����Ϊa g������������ת�����ǣ�
���ú���ĸ�Ĵ���ʽ��ʾ������ɲ�����
��5������Ӵ�����ͨ��2 molSO2(g)��1molO2(g)����һ���¶��£���Ӧ�ﵽƽ��ʱ��ýӴ����ڵ�ѹǿΪ��ʼʱ��0.75������SO2��ת����Ϊ ����ͬ�����£�����ʼʱͬʱͨ��a mol SO2��b mol O2��c mol SO3(g)ʱ���ﵽƽ��ʱ��ԭƽ���Ч������ʼʱά�ֻ�ѧ��Ӧ���淴Ӧ������У���c��ȡֵ��ΧΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012��ӱ�ʡ�����и�����ѧ����ĩ���п��������ۺϻ�ѧ�Ծ��������棩 ���ͣ�ʵ����
(15�֣�ͨ������һ��������Fe��Al�Ͻ���M����Һ��Ӧ����H2�ڱ�״���µ���������Լ�����Ͻ���Fe��Al������������������ͼװ������ȡH2,��������в�������ֱ������H2��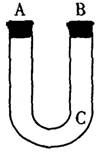
(1)C�����벣��˿���ţ�
(2) A�� ___________________________
(3) B��_____________________________
(4) ____________________________________
��5�� ____________________________________��
(6) �ڷ�Һ�¶��м���M����Һ��
(7) ______________________________________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010������������ѧ������ѧ�ڵ��ߴ��¿������ۣ���ѧ���� ���ͣ�ʵ����
(15��)������ͼ��ʾװ�ü�����ҩƷ̽����ҵ������ʱ���������ת���ʡ���֪C�к�����n mol�����������C��Dʱ�ֱܷ���ȫ����ijһ�����壬�Һ���װ���ڿ����е�CO2��

��ش��������⣺
��1�������װ�õ��������Ҽ���ҩƷ��ʼ����ʱ����Ӧ���еIJ�����
��
��2��ͨ���۲������ݵ��ݳ����ʿɿ���SO2��O2������������ȣ�Aװ�û���������� ��
��3��Ϊ����߲ⶨSO2ת���ʵ�ȷ�ȣ����в�����Ľ����е��ǣ�����ţ� ��
��Ϩ��ƾ��ƺ�ֹͣͨ��SO2������ͨ��O2һ��ʱ��
��Ϩ��ƾ��ƺ�ֹͣͨ��O2������ͨ��SO2һ��ʱ��
����C��D֮���װһ��ʢ��Ũ�����ϴ��ƿ
��4�������徭����֣�ʵ�������ϡ��װ��C�е���Һ���������м���������BaCl2��Һ���õ��ij�������Ϊw g����װ��D���ӵ�����Ϊa g������������ת�����ǣ�
���ú���ĸ�Ĵ���ʽ��ʾ������ɲ�����
��5������Ӵ�����ͨ��2 molSO2(g)��1molO2(g)����һ���¶��£���Ӧ�ﵽƽ��ʱ��ýӴ����ڵ�ѹǿΪ��ʼʱ��0.75������SO2��ת����Ϊ ����ͬ�����£�����ʼʱͬʱͨ��a mol SO2��b mol O2��c mol SO3(g)ʱ���ﵽƽ��ʱ��ԭƽ���Ч������ʼʱά�ֻ�ѧ��Ӧ���淴Ӧ������У���c��ȡֵ��ΧΪ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com