
��16�֣��۲����в���ͼ�Σ�����Ҫ��ش���������
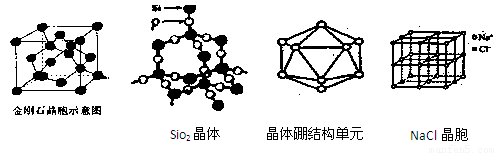
(1)�ɽ��ʯ����ṹ�;���ͼ֪�����ʯ��ԭ�Ӿ��壬������̼ԭ��ȡ
�ӻ�����γɦҼ���ÿ�������к�̼ԭ����Ϊ ����
(2)�����ӣ�P4���м���Ϊ �����ӵĿռ�ṹΪ ��ÿ1mol�����Ӻ� mol P-P���ۼ�������1���Ӱ���������P-P����������һ����ԭ������������ķ���ʽΪ ����ÿ��Pԭ�ӵŶԵ���������ԭ����λ���Ϳɵõ�����һ�������� �������ʽ����
(3)��SiO2����ṹ֪SiO2������ ���壬ÿ1mol SiO2���庬 mol Si-O���ۼ���
(4)��֪CaC2����ľ����ṹ��NaCl�������ƣ�����ͼ����CaC2������������C22-�Ĵ��ڣ�ʹ������һ��������������CaC2������Ca2+��λ����C.N��Ϊ ��C22-��O22+��Ϊ�ȵ����壬O22+�ĵ���ʽΪ ��1molO22+�к��м���ĿΪ ��

(5)������Ļ����ṹ��Ԫ��������ԭ����ɵ�����ʮ�����ԭ�Ӿ��塣���к���20���ȱ������κ�һ����Ŀ�Ķ��ǣ�ÿ�����Ǹ���һ��ԭ�ӣ��۲�ͼ�λش���������ṹ��Ԫ�� ����ԭ����ɣ������� ��B-B����
(6)�����mg NaCl��������ΪVcm3,��֪NaCl�����У����������Na+��Cl-��ľ���Ϊa cm,��ӵ�����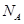 �ı���ʽΪ ��
�ı���ʽΪ ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��16�֣��۲����в���ͼ�Σ�����Ҫ��ش���������
(1)�ɽ��ʯ����ṹ�;���ͼ֪�����ʯ��ԭ�Ӿ��壬������̼ԭ��ȡ
�ӻ�����γɦҼ���ÿ�������к�̼ԭ����Ϊ ����
(2)�����ӣ�P4���м���Ϊ �����ӵĿռ�ṹΪ ��ÿ1mol�����Ӻ� mol P-P���ۼ�������1���Ӱ���������P-P����������һ����ԭ������������ķ���ʽΪ ����ÿ��Pԭ�ӵŶԵ���������ԭ����λ���Ϳɵõ�����һ�������� �������ʽ����
(3)��SiO2����ṹ֪SiO2������ ���壬ÿ1mol SiO2���庬 mol Si-O���ۼ���
(4)��֪CaC2����ľ����ṹ��NaCl�������ƣ�����ͼ����CaC2������������C22-�Ĵ��ڣ�ʹ������һ��������������CaC2������Ca2+��λ����C.N��Ϊ ��C22-��O22+��Ϊ�ȵ����壬O22+�ĵ���ʽΪ ��1molO22+�к��м���ĿΪ ��
(5)������Ļ����ṹ��Ԫ��������ԭ����ɵ�����ʮ�����ԭ�Ӿ��塣���к���20���ȱ������κ�һ����Ŀ�Ķ��ǣ�ÿ�����Ǹ���һ��ԭ�ӣ��۲�ͼ�λش���������ṹ��Ԫ�� ����ԭ����ɣ������� ��B-B����
(6)�����mg NaCl��������ΪVcm3,��֪NaCl�����У����������Na+��Cl-��ľ���Ϊa cm,��ӵ�����![]() �ı���ʽΪ ��
�ı���ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ���Ĵ�ʡ�����������и߶�������ĩ������ѧ�Ծ� ���ͣ������
��16�֣��۲����в���ͼ�Σ�����Ҫ��ش���������
(1)�ɽ��ʯ����ṹ�;���ͼ֪�����ʯ��ԭ�Ӿ��壬������̼ԭ��ȡ
�ӻ�����γɦҼ���ÿ�������к�̼ԭ����Ϊ ����
(2)�����ӣ�P4���м���Ϊ �����ӵĿռ�ṹΪ ��ÿ1mol�����Ӻ� mol P-P���ۼ�������1���Ӱ���������P-P����������һ����ԭ������������ķ���ʽΪ ����ÿ��Pԭ�ӵŶԵ���������ԭ����λ���Ϳɵõ�����һ�������� �������ʽ����
(3)��SiO2����ṹ֪SiO2������ ���壬ÿ1mol SiO2���庬 mol Si-O���ۼ���
(4)��֪CaC2����ľ����ṹ��NaCl�������ƣ�����ͼ����CaC2������������C22-�Ĵ��ڣ�ʹ������һ��������������CaC2������Ca2+��λ����C.N��Ϊ ��C22-��O22+��Ϊ�ȵ����壬O22+�ĵ���ʽΪ ��1molO22+�к��м���ĿΪ ��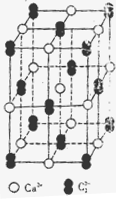
(5)������Ļ����ṹ��Ԫ��������ԭ����ɵ�����ʮ�����ԭ�Ӿ��塣���к���20���ȱ������κ�һ����Ŀ�Ķ��ǣ�ÿ�����Ǹ���һ��ԭ�ӣ��۲�ͼ�λش���������ṹ��Ԫ�� ����ԭ����ɣ������� ��B-B����
(6)�����mg NaCl��������ΪVcm3,��֪NaCl�����У����������Na+��Cl-��ľ���Ϊa cm,��ӵ�����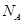 �ı���ʽΪ ��
�ı���ʽΪ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com