
��1����ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽��������___ ___________��

��2��Ϊ��ֹ����β����Ⱦ������������ˮ�����Ե����ʣ� ���� ��Һ���ն����������ԭ���ǣ��û�ѧ����ʽ��ʾ��_________��������һԭ������ҵ�ϳ������۵�ʯ�������չ�ҵ����β���Ƶ�Ư�ۣ�Ư�۵���Ч�ɷ���__ ____���ѧʽ��
��3���ݡ���������������2004��4��15����16�գ�λ�������н���������ԭ�����ܳ����
��������й©�ͱ�ը�¹ʡ��������߷���ʱ������9�����¹���ʧ��������3�����ˣ�15���˱�������ɢ�����������뱬ը�ֳ�ʱ�������ý���һ��Ũ�ȵ�ij�����ʵ�ˮ��Һ��ë����ס���ӣ������˲��õĸ������� ��
A��NaOH B��NaCl C��KBr D��Na2CO3
��6�֣���1����ʪ��ɫ������ɫ��������ɫ��������ɫ 1��
��2���������ƣ�NaOH����1��;2NaOH+Cl2=NaClO+NaCl+H2O 2�֣�Ca(ClO)2 1�� ��3��D 1��
��������
�����������1�����������Ư���ԣ����Գ�ʪ��ɫ������ɫ��������ɫ��������ɫ��
��2����ˮ�����ԣ����Կ���������������Һ���ն������������Ӧ�Ļ�ѧ����ʽ��2NaOH+Cl2��NaClO+NaCl+H2O��Ư�����ڻ�������Ч�ɷ��Ǵ�����ơ�
��3���������ƾ��и�ʴ�ԣ�A����ȷ���Ȼ��Ʋ��������������廯�������������������˵����壬�������ж���������ȷ�Ĵ�ѡD��
���㣺�������������ʡ�Ư�۵��Ʊ��ͳɷ֣��Լ�����й¶�¹ʴ�����
�������������е��Ѷȵ����⣬����ע�ػ��������ؿ���ѧ��������������������ѧ������˼ά�����ͷ�ɢ˼ά������Ҳ����������ѧ���Ĺ淶��������������Ĺؼ���������ס�������ʵĻ�ѧ���ʡ�


 Ӧ�����������Ĵ���ѧ������ϵ�д�
Ӧ�����������Ĵ���ѧ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��1����ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽��������
��1����ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽���������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��1����ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽��������
��1����ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽���������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��1����ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽��������
��1����ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽���������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
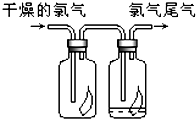 ��1����ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽������
��1����ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽�������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
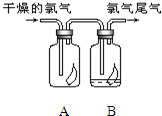 ��1����ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽��������
��1����ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽���������鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com