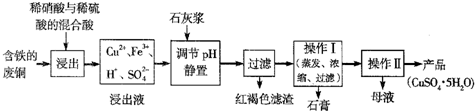
ij�Ͻ���м����Ҫ�ɷ�ΪCu��Fe��Al��������������ͭ��[Cu
2��OH��
2CO
3]����������������������������������Ͻ���м��ȡ������CuSO
4?5H
2O������ˮAlCl
3������Ĺ�����ͼ��ʾ��

��֪����Al
3+��Al��OH��
3��AlO
2-������ת����ϵ��Al
3+Al��OH��
3AlO
2-��̼����ܽ�Al��OH��
3��
��ش�
��1���ڷϽ���м��ĩ�м����Լ�A����������1�ķ�Ӧ�����ӷ���ʽ��
2Al+2OH-+2H2O=2 AlO2-+3H2��
2Al+2OH-+2H2O=2 AlO2-+3H2��
��
��2����Һ2�к��еĽ�����������
Fe2+
Fe2+
������2�ijɷ���
CO2��H2
CO2��H2
��
��3����Һ2ת��Ϊ����3�ķ�Ӧ�����ӷ���ʽ��
4Fe2++8OH-+O2+2H2O=4Fe��OH��3��
4Fe2++8OH-+O2+2H2O=4Fe��OH��3��
��
��4������2��ȡCuSO
4��Һ�ж��ַ�����
���ڹ���2�м���ŨH
2SO
4�������ȣ�ʹ����2ȫ���ܽ��CuSO
4��Һ����Ӧ�Ļ�ѧ����ʽ��
Cu+2H
2SO
4��Ũ��
CuSO
4+SO
2��+2H
2O
Cu+2H
2SO
4��Ũ��
CuSO
4+SO
2��+2H
2O
��
���ڹ���2�м���ϡH
2SO
4 ��ͨ��O
2�����ȣ�ʹ����2ȫ���ܽ��CuSO
4��Һ����Ӧ�����ӷ���ʽ��
��
��5����Һ1ת��Ϊ��Һ4�����У�������Һ1��ֱ�Ӽ����Լ�C��������
������Һ1��ֱ�Ӽ������ᣬ��ʹ��Һ4�л����Լ�A�е������ӣ���Na+�ȣ��������Ǵ�����AlCl3��Һ
������Һ1��ֱ�Ӽ������ᣬ��ʹ��Һ4�л����Լ�A�е������ӣ���Na+�ȣ��������Ǵ�����AlCl3��Һ
��
��6��ֱ�Ӽ���AlCl
3?6H
2O���ܵõ���ˮAlCl
3��SOCl
2Ϊ��ɫҺ�壬������ˮ��Ӧ����HCl��һ�־���Ư���Ե����壮AlCl
3?6H
2O��SOCl
2��ϼ�����ȡ��ˮAlCl
3����Ӧ�Ļ�ѧ����ʽ��
AlCl
3?6H
2O+6SOCl
2 AlCl
3+12HCl��+6SO
2��
AlCl
3?6H
2O+6SOCl
2 AlCl
3+12HCl��+6SO
2��
��