
 ��ش��������⣺
��ش��������⣺
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ʹpH��ֽ������Һ��NH4+Na+SO42�� Fe2�� |
| B��������ˮ�У�Cl��NO3��Na+SO32�� |
| C��pH=1����Һ�У�K+MnO4��Cl�� NH4�� |
| D��FeCl3��Һ�У�K��Na�� SO42�� AlO2�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��Zn2+��Cu��Fe3+ | B��Zn��Cu2+��Fe3+ |
| C��Zn2+��Cu��Cu2+��Fe2+ | D��Zn2+��Cu2+��Fe��Fe2+ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A�����д���NO��2��ˮ��Һ�У�Na+��Fe2+��SO2-4��H+ |
| B����pH=12����Һ�У�ClO����S2-��Na+��K+ |
| C����ʹpH��ֽ��������Һ NH4+Na+SO42��Fe3�� |
| D�����д���HCO��3�ij�������Һ�У�K+��C6H5O����Cl����CH3COO�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��Na����Al3����Cl����OH����5��2��7��4�� | B��NH4+��Ba2����OH����Cl����1��1��1��2�� |
| C��Fe3����Na����Cl����S2����1��2��3��1�� | D��K����H����I����HCO3����1��1��1��1�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��Mg2����H����C1����OH���������� | B��Na����Ba2����CO ��NO ��NO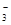 |
C��Na����H����Cl����CO ������ ������ | D��K����Cu2����NO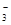 ��SO ��SO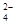 �� �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��HCO3����Al3+��Na+��SO42�� | B��I����NO3����K+��NH4+ |
| C��MnO4����Cl����SO42����K+ | D��SiO32����SO32����Na+��Cl�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��������Һ��Fe2+��NO3����SO42����Na+ |
| B��������Һ��S2����K+��HCO3����AlO2�� |
| C�����Ի������Һ��Na����K����Cl����SO42�� |
| D���������۷ų���������Һ��Na+��Cl����NH4+��NO3�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��K����HCO3-��Na+��SO | B��Na����Cu2����SO��Cl�� |
| C��Ba2����NO3����Cl����K�� | D��Ca2����K����CO��NO |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com