
��13�֣���������������ѭ��ѧԭ������������й����������̻ش����⡣
��1��ij��ѧ����С���Ժ���Ϊԭ�ϻ�������⣬���������ͼ��ʾ��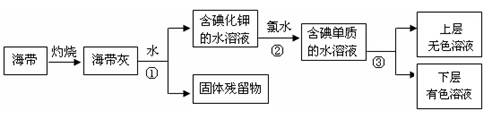
�����ٵ������� ��������ʹ�õ��Լ���д��ѧʽ�� ���������з�����Ӧ�����ӷ���ʽΪ ��
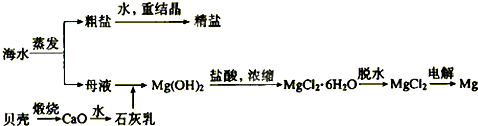
��2����ˮ���ۺ����ÿ����Ʊ������ƺ�þ������������ͼ��ʾ��
����������������Mg(OH)2���������ӷ���ʽΪ������ ��������Mg(OH)2���������ᷴӦ�����ӷ���ʽΪ ��
��ʵ�����ォ�����Ƴɾ��εĹ����У����ܽ⡢���ˡ�������������IJ����ж�Ҫ�õ����������ֱ�˵�������������ʹ�ò����������ã�
�ܽ�ʱ��____________������ʱ��____________������ʱ��______________��
�۹�ҵ�ϰѵ�ⱥ��ʳ��ˮ��Ϊ���ȼҵ����������ɵ�ⱥ��ʳ��ˮ�Ļ�ѧ����ʽ�� ��NaCl+�� ��H2O  �� ��NaOH+H2��+�� ���� ����
�� ��NaOH+H2��+�� ���� ����

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��13�֣���������������ѭ��ѧԭ������������й����������̻ش����⡣
��1��ij��ѧ����С���Ժ���Ϊԭ�ϻ�������⣬���������ͼ��ʾ��
�����ٵ������� ��������ʹ�õ��Լ���д��ѧʽ�� �������� �з�����Ӧ�����ӷ���ʽΪ ��
��2����ˮ���ۺ����ÿ����Ʊ������ƺ�þ������������ͼ��ʾ��
����������������Mg(OH)2���������ӷ���ʽΪ������ ��������Mg(OH)2���������ᷴӦ�����ӷ���ʽΪ ��
��ʵ�����ォ�����Ƴɾ��εĹ����У����ܽ⡢���ˡ�������������IJ����ж�Ҫ�õ����������ֱ�˵�������������ʹ�ò����������ã�
�ܽ�ʱ��____________������ʱ��____________������ʱ��______________��
�۹�ҵ�ϰѵ�ⱥ��ʳ��ˮ��Ϊ���ȼҵ����������ɵ�ⱥ��ʳ��ˮ�Ļ�ѧ����ʽ �� ��NaCl+�� ��H2O�� ��NaOH+H2��+�� ���� ����
![]()
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�츣��ʡ��һ��һѧ���������⿼�Ի�ѧ�Ծ� ���ͣ������
��13�֣���������������ѭ��ѧԭ������������й����������̻ش����⡣
��1��ij��ѧ����С���Ժ���Ϊԭ�ϻ�������⣬���������ͼ��ʾ��
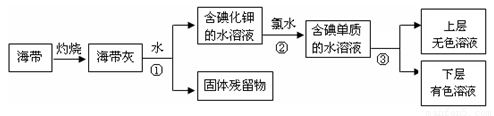
�����ٵ������� ��������ʹ�õ��Լ���д��ѧʽ�� �������� �з�����Ӧ�����ӷ���ʽΪ ��
��2����ˮ���ۺ����ÿ����Ʊ������ƺ�þ������������ͼ��ʾ��
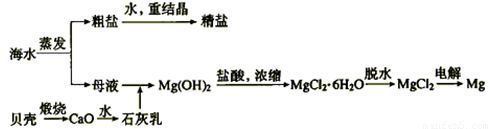
����������������Mg(OH)2���������ӷ���ʽΪ������ ��������Mg(OH)2���������ᷴӦ�����ӷ���ʽΪ ��
��ʵ�����ォ�����Ƴɾ��εĹ����У����ܽ⡢���ˡ�������������IJ����ж�Ҫ�õ����������ֱ�˵�������������ʹ�ò����������ã�
�ܽ�ʱ��____________������ʱ��____________������ʱ��______________��
�۹�ҵ�ϰѵ�ⱥ��ʳ��ˮ��Ϊ���ȼҵ����������ɵ�ⱥ��ʳ��ˮ�Ļ�ѧ����ʽ �� ��NaCl+�� ��H2O
 �� ��NaOH+H2��+�� ���� ����
�� ��NaOH+H2��+�� ���� ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�츣��ʡ�����и�һ��ѧ���������⿼�Ի�ѧ�Ծ� ���ͣ�ʵ����
��13�֣���������������ѭ��ѧԭ������������й����������̻ش����⡣
��1��ij��ѧ����С���Ժ���Ϊԭ�ϻ�������⣬���������ͼ��ʾ��
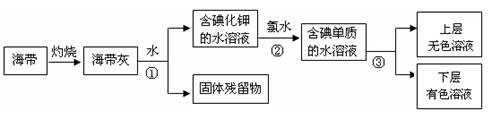
�����ٵ������� ��������ʹ�õ��Լ���д��ѧʽ�� �������� �з�����Ӧ�����ӷ���ʽΪ ��
��2����ˮ���ۺ����ÿ����Ʊ������ƺ�þ������������ͼ��ʾ��

����������������Mg(OH)2���������ӷ���ʽΪ������ ��������Mg(OH)2���������ᷴӦ�����ӷ���ʽΪ ��
��ʵ�����ォ�����Ƴɾ��εĹ����У����ܽ⡢���ˡ�������������IJ����ж�Ҫ�õ����������ֱ�˵�������������ʹ�ò����������ã�
�ܽ�ʱ��____________������ʱ��____________������ʱ��______________��
�۹�ҵ�ϰѵ�ⱥ��ʳ��ˮ��Ϊ���ȼҵ����������ɵ�ⱥ��ʳ��ˮ�Ļ�ѧ����ʽ �� ��NaCl+�� ��H2O  �� ��NaOH+H2��+�� ���� ����
�� ��NaOH+H2��+�� ���� ����
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com