
������һ����Ҫ�Ļ���ԭ�ϣ��㷺����ҩ���������߷��Ӻϳɵȹ�ҵ�����ᾧ�����ȵ�100��ʱʧȥ�ᾧˮ����Ϊ��ˮ���ᡣijѧϰС���ͬѧ���Ը�����Ϊԭ����ˮ�⡪������ˮ��ѭ��������ȡ���ᡣ
|
|
 �����������Ϣ�ش��������⣺
�����������Ϣ�ش��������⣺��������ƿ��2�֣���70�棨1�֣������ڿ����¶ȣ�ʹװ�����Ⱦ��ȣ�2�֣�
��BC��2�֣��Ȣ���ʽ�ζ��ܣ�1�֣� ��CD��2�֣�������ƿ�е������һ�θ�����ر�Һ����ƿ����Һ���dz��ɫ�Ұ���Ӻ�ɫ��2�֣� ��87.5%��2�֣�
���������������1������װ�õĽṹ�ص��֪��AӦ����������ƿ��
��2������ͼ2��֪�����¶�Ϊ70��ʱ�������������ߣ�����ѡ�����ѷ�Ӧ�¶���70�档�����¶Ȳ�����100�棬���Բ���ˮԡ���ȣ���������ˮԡ����ʱ���ڿ����¶ȣ�����ʹװ�����Ⱦ��ȡ�
��3��A����ʵ������У�ͨ��������ȴ������Һ���ò����ϴ�ľ��������Ӧ������Ȼ��ȴ��A����ȷ��B����ϴ�ӳ���ʱ��Ӧ��Сˮ��ͷ��ʹϴ�Ӽ�����ͨ���������ֹ�����ܽ�������ʧ��B��ȷ��C������װ���д���ѹǿ�����Ϊ�˼���ϴ���Ƿ���ȫ��Ӧ��������ƿ�밲ȫƿ֮����Ƥ�ܣ�������ƿ�Ͽڵ���������Һ���Թ��н������ʵ�飬C��ȷ��D����������ֱ�Ӽ��ȣ�����ʹ���ᾧ��ֽ⣬D����ȷ����ѡBC��
��4���ٲ�����Һ�����ԣ�������ȡ25.00mL������Һ����������ʽ�ζ��ܡ�
��A����ϴ�ζ���ʱ��Ӧ�ӵζ����Ͽڼ��������������Һ��ʹ�ζ����ڱڳ����ϴ��������Ҫ������A����ȷ��B����Һ��ȡ������Һʱ�����ܽ����촦��Һ�崵����ƿ������ʹʵ�����ƫ�ߣ�B����ȷ��C���ζ�ʱ�������������ڿ�ס���������Ŀ���������������ʹ���ɶ�©����Һ��C��ȷ��D���ζ��յ��ȡ�ζ��̶ܿ�ʱ�����ӱ�ҺҺ�棬���ڶ������϶������������Զ���ƫ��ʹʵ�����ƫ�ߣ�D��ȷ����ѡCD��
���������Ը��������Һ���Ϻ�ɫ�����Եζ��յ���ж�����������ƿ�е������һ�θ�����ر�Һ����ƿ����Һ���dz��ɫ�Ұ���Ӻ�ɫ��
�ܷ�Ӧ�����ĸ�����ص����ʵ�����0.02L��0.1000mol/L��0.002000mol�����Ը��ݷ���ʽ5H2C2O4+2MnO4��+6H+��2Mn2++10CO2��+8H2O��֪�����IJ�������ʵ�����0.005000mol�����ԭ��Ʒ�в�������ʵ�����0.005000mol��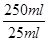 ��0.05000mol/L����������0.05000mol/L��126g/mol��6.3g�����Բ���Ĵ�����
��0.05000mol/L����������0.05000mol/L��126g/mol��6.3g�����Բ���Ĵ�����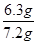 ��100%��87.5%��
��100%��87.5%��
���㣺��������ʶ�������Ʊ���ʵ��̽�����ζ�ʵ����й��жϺͼ���



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����п��ĩ�㷺Ӧ������Ϳ�ϡ��մɡ�������ҽҩ����������ҵ��Ϊ�ۺ�Ӧ����Դ������ұ��п��п��Ʒ�ӹ���ҵ���յ�п��������ZnO��FeO��Fe2O3��CuO��Al2O3�����ʣ�����ȡ����п��������ͼ��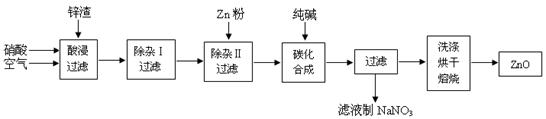
�й��������������ȫ��pH���±���
��1������������У�����п����ɷ�ĩ��ͨ�������ͬʱ����������������� ��
��2���������նദ�漰�����ˡ���ʵ�����й�����Ҫʹ�õIJ����������ձ��� ��
��3�����ڡ����Ӣ��У���������KMnO4��Һ����Ŀ���� ��KMnO4�Ǹ÷�Ӧ�� ������������ԭ����������Һ��pH����4��Ŀ���� ��
���ڡ�����II���У�����п�۵�Ŀ���� ��
��4���ڡ�̼���ϳɡ��У��������м�ʽ̼��п��Zn2(OH)2CO3�ݺ�CO2�ȣ���д���÷�Ӧ�Ļ�ѧ����ʽ ��
��5���������У����˷�������϶࣬�����Ե�ȱ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��15�֣�S2Cl2�����������л����Ȼ����Լ���ʵ���ҿ���������װ�����Ʊ�S2Cl2�������ּг���������ȥ��
��֪�����Ʊ��ķ�Ӧ����ʽΪ��
�ڷ�Ӧ�漰�ļ��������������£�
| ���� ���� | �۵� | �е� | �Ż�� | �������� |
| ��� | 119.2�� | 446.6�� | 363�� | / |
| ��� | 112.8�� | 446.6�� | 363�� | / |
| S2Cl2 | -77�� | 137�� | / | ��ˮǿ�ҷֽ�����S��SO2��HCl |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�����г����⣬����������Ԫ�أ���̼Ԫ�غ���Ԫ�ء�����̼��Ҫ��̼��������̬���ڣ���ʹ�������ܼ�Ӳ���࣬������������;����һ���������ֵ�ԭ�ϡ�ij��ȤС����ư���ͼ��ʾ��ʵ��װ�ã��ⶨ�����еĺ�̼����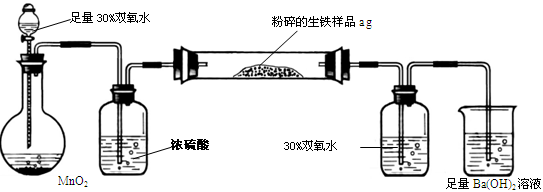
A B C D E
��ش��������⣺
��1���������������к�Ԫ�أ���ʹ���������ȴ��ԡ���Ԫ�������������п��ܴ��ڵļ�̬��
| A����2���� | B��0�� �� | C��+4���� | D��+6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
PCl3�����ڰ뵼�����������ӡ���ɢ�����й����ʵIJ����������£�
| | �۵�/�� | �е�/�� | �ܶ�/g��mL��1 | ���� |
| ���� | 44��1 | 280��5 | 1��82 | 2P(����)+3Cl2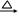 2PCl3��2P+5Cl2(����) 2PCl3��2P+5Cl2(����) 2PCl5 2PCl5 |
| PCl3 | ��112 | 75��5 | 1��574 | ��ˮ����H3PO3��HCl����O2����POCl3 |
| POCl3 | 2 | 105��3 | 1��675 | ��ˮ����H3PO4��HCl��������PCl3 |
 H3PO4+2HI��b��(NH4)3BO3��Һ����HI��H3PO4��Ӧ����H3BO3�����ᣩ��c��Na2S2O3��Һ�ɶ����ⶨ�⣺I2+2Na2S2O3��Na2S4O6+2NaI
H3PO4+2HI��b��(NH4)3BO3��Һ����HI��H3PO4��Ӧ����H3BO3�����ᣩ��c��Na2S2O3��Һ�ɶ����ⶨ�⣺I2+2Na2S2O3��Na2S4O6+2NaI�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
KMnO4��Һ������������ԭ��Ӧ�ζ��ı�Һ�仹ԭ����ΪMn2+������KMnO4��ǿ�����ԣ�������Һ�����ױ������л�ˮ�е�ijЩ������ԭ�����ʻ�ԭ����������������MnO(OH)2���������KMnO4����Һ�IJ���������ʾ��
�ٳ�ȡ�Զ�����������KMnO4��������ˮ�У�����Һ���Ȳ�������1 h�������ײ���©�����˳�ȥ���ܵ�MnO(OH)2���۹��˵õ�KMnO4��Һ�����棻������������ԭ�ζ���������70��80 ���������û��Լ�(���ȸߡ���Է��������ϴ��ȶ��ԽϺõ�����)��Һ�궨��Ũ�ȡ�
��ش��������⣺
(1)���ƺõ�KMnO4��Һ���淽���� ��ȷ��ȡһ�������KMnO4��Һ��Ҫʹ�õ�������____________��
(2)�����������У����ڱ궨KMnO4��Һ�Ļ��Լ����ѡ��________(�����)��
| A��H2C2O4��2H2O | B��FeSO4 | C��Ũ���� | D��Na2SO3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��14�֣��̷����壨FeSO4��7H2O��M=278g/mol��������ȱ����ƶѪҩƷ����Ҫ�ɷ֡�ʵ�����������᳧����������Ҫ�ɷ�ΪFe2O3������FeS��SiO2�����Ʊ��̷��Ĺ������£�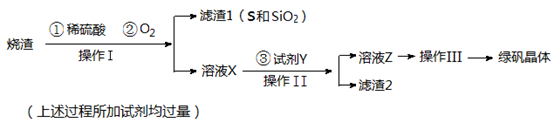
�Իش�
��1��������Ϊ ����д�������ƣ���
��2���Լ�Y����ҺX��Ӧ�����ӷ���ʽΪ ��
��3���������˳������Ϊ�� ����ȴ�ᾧ������ �� �����
��4��ijͬѧ������KMnO4��Һ�ⶨ�̷���Ʒ��Fe2+������
a.��ȡ11.5g�̷���Ʒ���ܽ⣬���Ƴ�1000mL��Һ��b.��ȡ25.00mL������Һ����ƿ�У�
c.�������ữ��0.01000mol/L KMnO4��Һ�ζ����յ㣬����KMnO4��Һ�����ƽ��ֵΪ20.00mL��
�ٲ���a������Һʱ��Ҫ�IJ�������������������Ͳ���ձ�����ͷ�ι��⣬����
�ڸ�ͬѧ��Ƶ����еζ���ʽ����������� (�гֲ�����ȥ)(����ĸ���)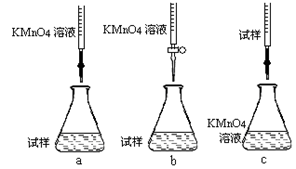
�۵ζ�ʱ������Ӧ�����ӷ���ʽΪ�� ���������������� ����������
���жϴ˵ζ�ʵ��ﵽ�յ�ķ����� �����ڵ��յ��ȡ�ζ��̶ܿ�ʱ������KMnO4��ҺҺ�棬������������ȷ����ʹ�ⶨ��� ���ƫ�ߡ���ƫ�͡�����Ӱ�족����
�ݼ���������Ʒ��FeSO4��7H2O����������Ϊ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Ϊ�ⶨ��������茶��塾��NH4��2Fe (SO4)2 �� xH2O�������ĺ�����ijʵ��С����������ʵ�飺
����һ���õ�����ƽȷ����5.000g��������茶��壬���Ƴ�250ml��Һ��
�������ȡ������Һ25.00ml����ƿ�У���ϡH2SO4�ữ����0.010mol/L KMnO4��Һ�ζ���Fe2+ǡ��ȫ��������Fe3+��ͬʱ��MnO4-����ԭ��Mn2+��
���ظ���������Ρ�
��ش��������⣺
��1�����������������Һ�IJ������������ǣ������� ��ת�ơ�ϴ�Ӳ�ת�ơ� ��ҡ�ȡ�
��2���� �ζ���ʢ��KMnO4��Һ��
��3�����������һ��KMnO4��Һ,���� ,������ζ��յ㡣��Ӧ�����ӷ���ʽ��
��4���ζ�������±���ʾ��
| �ζ����� | ������Һ�����/mL | ����Һ����� | |
| �ζ�ǰ�̶�/mL | �ζ���̶�/mL | ||
| 1 | 25.00 | 1.05 | 21.04 |
| 2 | 25.00 | 1.50 | 24.50 |
| 3 | 25.00 | 0.20 | 20.21 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����ÿ���и����������ʣ����Ƕ����÷�Һ©���������
| A���屽��ˮ���ƾ���ˮ | B�����Ȼ�̼��ˮ���屽�ͼ��� |
| C�����ͺ�ˮ�������屽 | D�����ͺ�ˮ�������ˮ |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com