


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

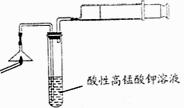
| A���������� | B������ʱ���� |
| C������ʱ���ʺ�����λ�÷��ˣ���Ҫ���룩 | D������ƿ�ô�װҺ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

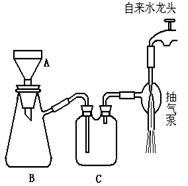
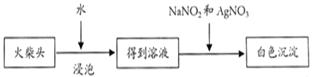
|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
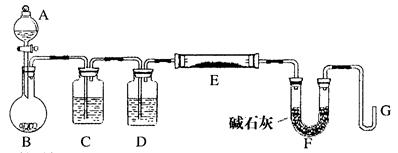
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A������ �����������ۣ���������ȩ�� �����������ۣ���������ȩ�� |
B�� ij±�������� ij±��������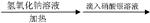 ���յij������ǰ�ɫ�� ���յij������ǰ�ɫ�����ۣ���±�����в�����ԭ�� |
C��ij ��Һ ��Һ ð�Ű��� ð�Ű��� �����������̣� �����������̣����ۣ�����ҺΪŨ���� |
D����ɫ��Һ �ʻ�ɫ�����ۣ�����Һһ��������Ԫ�� �ʻ�ɫ�����ۣ�����Һһ��������Ԫ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
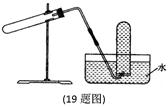
| A������CaCO3��CO2 |
| B����Cu��ϡHNO3��Ӧ��NO |
| C����NH4Cl��ŨNaOH��Һ��Ӧ��NH3 |
| D����NaCl��ŨH2SO4��Ӧ��HC1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������ʱ��ǿ�����ɣ�����ͭ������ֺ�ɫ���� |
| B�������ڸ����������Ȼ��ƹ��� |
| C������ʱ����������δ��ȫ���ֹͣ���� |
| D�����ȵ���������ڿ�������ȴ���� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com