
 O2��g��=H2O��l����H=-285.8kJ/mol
O2��g��=H2O��l����H=-285.8kJ/mol  O2��g��=CO2��g����H=-282.6kJ/mol
O2��g��=CO2��g����H=-282.6kJ/mol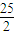 O2��g��=8CO2��g��+9H2O��l����H=-5472kJ/mol
O2��g��=8CO2��g��+9H2O��l����H=-5472kJ/mol

 �������Ӧ���⼯ѵϵ�д�
�������Ӧ���⼯ѵϵ�д� �ۺ��Բ�ϵ�д�
�ۺ��Բ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| 1 |
| 2 |
| 1 |
| 2 |
| 25 |
| 2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| 1 |
| 2 |
| 1 |
| 2 |
| 25 |
| 2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| 1 |
| 2 |
| 1 |
| 2 |
| 25 |
| 2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������һ����̼�����顢������Ȼ�ѧ����ʽ�ֱ�Ϊ
H2(g)+1/2O2(g)====H2O(l);
��H=-285.8 kJ��mol-1
CO(g)+1/2O2(g)====CO2(g);
��H=-283.0 kJ��mol-1
C8H18(l)+25/2O2(g)====8CO2(g)+9H2O(l);
��H=-5 518 kJ/mol
CH4(g)+2O2(g)====CO2(g)+2H2O(l);
��H=-890.3 kJ��mol-1
��ͬ������H2��CO��C8H18��CH4��ȫȼ��ʱ���ų��������ٵ���( )
A.H2(g) B.CO(g) C.C8H18(l) D.CH4(g)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012������ʡ�߶���ѧ�����п��Ի�ѧ�Ծ� ���ͣ�ѡ����
������һ����̼�����顢������Ȼ�ѧ����ʽ�ֱ�Ϊ��
H2(g)��1/2O2(g)��H2O(l)�� ��H����285.8 kJ / mol
CO(g)��1/2O2(g)��CO2(g)�� ��H����283.0 kJ / mol
C8H18(l)��25/2O2(g)��8CO2(g)��9H2O(l)�� ��H����5518 kJ / mol
CH4(g)��2O2(g)��CO2(g)��2H2O(l)�� ��H����890.3 kJ / mol
��ͬ������������һ����̼�����顢������ȫȼ��ʱ���ų��������ٵ���
A�� H2(g) B�� CO(g) C�� C8H18(l) D��CH4(g)
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com