
ijС��ͬѧ���о�SO2�����ʡ�
��1������صĺ������ʷ�Ϊ���±���ʾ��3�飬��2��������X�Ļ�ѧʽ������������
|
��1�� |
��2�� |
��3�� |
|
S�����ʣ� |
SO2��X��Na2SO3��NaHSO3 |
SO3��H2SO4��Na2SO4��NaHSO4 |
��2��������ͼ��ʾ��װ���о�SO2�����ʣ�
���۵㣺SO2��76.1�棬SO3 16.8�棻�е㣺SO2��10�棬SO3 45�棩
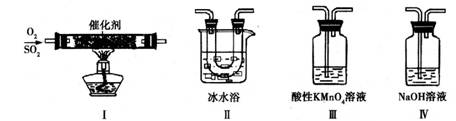
��װ�â�ģ�ҵ������SO2�������ķ�Ӧ���仯ѧ����ʽ��������������
�ڼ�ͬѧ������˳������װ�ã�װ�â������������������������
װ�â�����Һ����ɫ������Mn2+��ͬʱpH���ͣ���÷�Ӧ�����ӷ���ʽ��
����������������������������
����ͬѧ������˳������װ�ã���װ�â�����70mL 2mol��L��1NaOH��Һ����Ӧ������5.12g����װ�â��з�����Ӧ�Ļ�ѧ����ʽ������������������
 �Ͻ�ƽ��У����ϵ�д�
�Ͻ�ƽ��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ�����ʡ��ɳ�и����������¿���ѧ�Ծ��������棩 ���ͣ������
��1��ij��ѧ��ȤС���ͬѧ����Cl2��NH3���Ʊ������ʼ����ʵ������̺Ͳ���װ�����£�
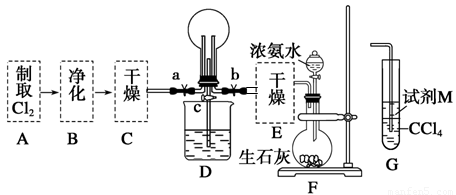
������A��Gװ�����һ����ʵ����֤Cl2��Fe3����I2��������ǿ��ΪCl2>Fe3��>I2(ʵ���в��ϵ�С����Gװ���е��Թ�)��A�з�Ӧ����KMnO4��Ũ���ᣬ��д��A�з�����Ӧ�Ļ�ѧ����ʽ�� ����д���Լ�MΪ ��Һ��֤��������ΪCl2>Fe3��>I2��ʵ�������� ��
����֪3Cl2��2NH3=6HCl��N2����D����ƿ�г�������ɫ����ر�a��c��b��D�е�����Ϊ����ɫ������ʧ���������̣���Ӧһ��ʱ��ر�b��c���۲쵽������Ϊ_________________________________________________________________��
��2��ij��ˮ�к���һ������Na+��SO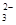 �����ܺ���CO
�����ܺ���CO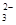 ��ij�о�С�����ⶨ����SO
��ij�о�С�����ⶨ����SO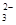 ��Ũ�ȣ��������ʵ�鷽����
��Ũ�ȣ��������ʵ�鷽����

�ٴ������Լ���ѡ���Լ�XΪ_________������ţ���
A��0.1 mol/L KMnO4(H2SO4�ữ)��Һ B��0.5 mol/L NaOH��Һ
C��������ˮ D��KI��Һ
�ڼ����Լ�X����SO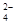 �����ӷ���ʽΪ_____________________________________��
�����ӷ���ʽΪ_____________________________________��
��֤���÷�ˮ���Ƿ���CO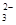 ��ʵ�鷽��Ϊ
��
��ʵ�鷽��Ϊ
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ�����ʡ��ɳ�и����������¿���ѧ�Ծ��������棩 ���ͣ������
��1��ij��ѧ��ȤС���ͬѧ����Cl2��NH3���Ʊ������ʼ����ʵ������̺Ͳ���װ�����£�
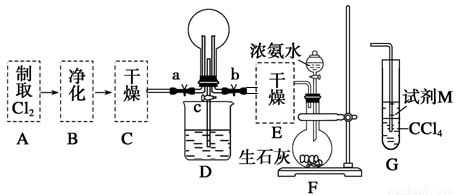
������A��Gװ�����һ����ʵ����֤Cl2��Fe3����I2��������ǿ��ΪCl2>Fe3��>I2(ʵ���в��ϵ�С����Gװ���е��Թ�)��A�з�Ӧ����KMnO4��Ũ���ᣬ��д��A�з�����Ӧ�Ļ�ѧ����ʽ�� ����д���Լ�MΪ ��Һ��֤��������ΪCl2>Fe3��>I2��ʵ�������� ��
����֪3Cl2��2NH3=6HCl��N2����D����ƿ�г�������ɫ����ر�a��c��b��D�е�����Ϊ����ɫ������ʧ���������̣���Ӧһ��ʱ��ر�b��c���۲쵽������Ϊ_________________________________________________________________��
��2��ij��ˮ�к���һ������Na+��SO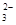 �����ܺ���CO
�����ܺ���CO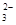 ��ij�о�С�����ⶨ����SO
��ij�о�С�����ⶨ����SO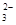 ��Ũ�ȣ��������ʵ�鷽����
��Ũ�ȣ��������ʵ�鷽����

�ٴ������Լ���ѡ���Լ�XΪ_________������ţ���
A��0.1 mol/L KMnO4(H2SO4�ữ)��Һ B��0.5 mol/L NaOH��Һ
C��������ˮ D��KI��Һ
�ڼ����Լ�X����SO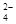 �����ӷ���ʽΪ_____________________________________��
�����ӷ���ʽΪ_____________________________________��
��֤���÷�ˮ���Ƿ���CO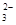 ��ʵ�鷽��Ϊ
��
��ʵ�鷽��Ϊ
��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com