
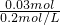 =0.15L=150mL��
=0.15L=150mL��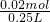 =0.08mol/L��
=0.08mol/L��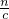 ���������������������Һ������ΪNaOH�������������غ��֪n��NaOH��=n��Na+�����ٸ���c=
���������������������Һ������ΪNaOH�������������غ��֪n��NaOH��=n��Na+�����ٸ���c=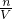 ���㣮
���㣮


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ˮ�� |
| ˮ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �¶�/�� | 25 | t1 | t2 |
| Kw/mol2?L-2 | 1��10-14 | a | 1��10-12 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�갲����ѧ�߶�������ϰ��ѧ�����ģ� ���ͣ�ʵ����
������ʵ��������ʵ����ʵ��������ȷ���� ������ţ���
�����Թ��еμ�Һ��ʱ��Ϊ��ʹҺ��ε��Թ���Ӧ����ͷ�ι������Թ��У�
��һС������Ƽ���ˮ�к�Ѹ���۳�С��ͣ����ˮ���ζ���������֨֨����������
������100mL1.00mol/L��NaCl��Һʱ������������ƽ��ȡ5.85g NaCl���壻
������ܺ���SO42����SO32������Һ�м�����������ᣬ�ټ���Ba(NO3)2��Һ���ɼ���SO42���Ĵ��ڣ�
������NaCl��Һ�Եõ�NaCl����ʱ�����ؽ���Һ���ɣ�
����100��ʱ��NaOHϡ��Һ�еμӱ��͵�FeCl3��Һ�����Ʊ�Fe(OH)3���壻
����ͼ���ɹ۲쵽���������Ƶ�ָ��ƫת��
����AlCl3��Һ�еμ�NaOH��Һ����NaOH��Һ�еμ�AlCl3�� Һ��������ͬ��
������0.1mol��L��1�Ĵ�����Һ����pH��ֽ�ⶨ����Һ��pH����
��ȷ�IJ�����
����Ϊ����ҺpH�ķ�Χһ������ ֮�䡣�������һ����ʵ�鷽��֤��������Һ�ʼ�������CO32������ģ� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com