
 ��D��Ԫ������������Ӧˮ������Ե�ǿ�����ѧʽ�� ��
��D��Ԫ������������Ӧˮ������Ե�ǿ�����ѧʽ�� �� ________________________________��
________________________________�� ��У��ʦ������ҵ���Ӻ����Ծ�ϵ�д�
��У��ʦ������ҵ���Ӻ����Ծ�ϵ�д� ȫ�̽��ϵ�д�
ȫ�̽��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
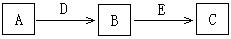
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
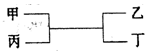
| A�������� | B��̼�� |
| C��þ��̼ | D���������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 D ��E��F�dz��������壬����A��B��C��DΪ���ʣ��йص�ת����ϵ����ͼ��ʾ(��Ӧ����������ȥ����
D ��E��F�dz��������壬����A��B��C��DΪ���ʣ��йص�ת����ϵ����ͼ��ʾ(��Ӧ����������ȥ����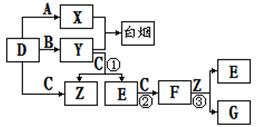
 �۵����ӷ���ʽΪ ��
�۵����ӷ���ʽΪ �� Ϊ ��
Ϊ �� ��Һ��ˮ�������c(H+)��c(OH-)�˻�Ϊ1��10-22
��Һ��ˮ�������c(H+)��c(OH-)�˻�Ϊ1��10-22�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 ZX2��Ӧ����һ�ֻ���
ZX2��Ӧ����һ�ֻ��� ��Y2ZX3����֪����Ԫ��ԭ�ӵĵ�������Ϊ25����Z��Y��ԭ������֮�ͱ�X��ԭ��������2������1��Zԭ�ӵ������������Ǵ�����������2�����Իش�
��Y2ZX3����֪����Ԫ��ԭ�ӵĵ�������Ϊ25����Z��Y��ԭ������֮�ͱ�X��ԭ��������2������1��Zԭ�ӵ������������Ǵ�����������2�����Իش� Z_________
Z_________�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
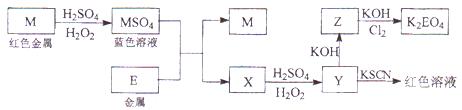
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 ���ˮ����֮������Ӧ�Ļ�ѧ����ʽ�� ��
���ˮ����֮������Ӧ�Ļ�ѧ����ʽ�� ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 ԭ����֮��Ϊ2��1�Ļ�����X��1��1�Ļ�����Y��D��BҲ�����ԭ����֮��Ϊ2:1�Ļ�����M��1:1�Ļ�����Z��1molC���ʸ�X��Ӧ�ų�0.5mol����B2,1molD���ʸ�X��Ӧ�ų�11.2L����������A2��1molZ�������X��Ӧ�ų�0.5mol����B2���Իش�
ԭ����֮��Ϊ2��1�Ļ�����X��1��1�Ļ�����Y��D��BҲ�����ԭ����֮��Ϊ2:1�Ļ�����M��1:1�Ļ�����Z��1molC���ʸ�X��Ӧ�ų�0.5mol����B2,1molD���ʸ�X��Ӧ�ų�11.2L����������A2��1molZ�������X��Ӧ�ų�0.5mol����B2���Իش��鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com