
ij��A 0.2 mol�������г��ȼ�պ����ɻ�����B��C��1.2 mol��������������⣺
(1)��A�ķ���ʽ��_________��
(2)��ȡһ��������A���ȼ�պ�����B��C��3 mol������_________g��A�μ��˷�Ӧ��ȼ��ʱ���ı�״���µ�����_________ L��
(3)����A����ʹ��ˮ��ɫ������һ��������������������ȡ����Ӧ����һ�ȴ���ֻ��һ�֣�����A�Ľṹ��ʽ��___________��
(4)����A��ʹ��ˮ��ɫ���ڴ�����������H2�����ӳɷ�Ӧ������2��2-�������飬����A��������_________���ṹ��ʽ��_________��
(5)��A������̼ԭ�ӵ�A��ϩ��ͬϵ���ͬ���칹�干��_________�֡�
(1)C6H12 (2)42 100.8 (3)
(4)3��3-����-1-��ϩ 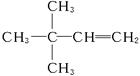 (5)3
(5)3
(1)�������������A�ķ���ʽΪC6H12��(3)C6H12������ϩ����Ҳ�����ǻ���������Ϊ��A����ʹ��ˮ��ɫ��˵����A���ڻ�����������C6H12Ϊ�����飬��ṹ��ʽΪ ��
��
(4)����A��ʹ��ˮ��ɫ��˵��C6H12Ϊϩ��������������Ϊ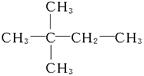 ������AӦΪ
������AӦΪ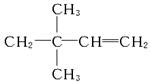 ������Ϊ3��3��-����-1-��ϩ��
������Ϊ3��3��-����-1-��ϩ��
(5)ͬ���칹���У���ϩ������
CH3��CH2��CH==CH2��CH3��CH==CH��CH3��![]() 3�֡�
3�֡�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(1)��A�ķ���ʽ____________��B��C�ķ���ʽ�ֱ���_____________��_____________��
(2)��ȡһ��������Aȼ�պ�����B��C��3 mol������___________g��A�μ��˷�Ӧ��ȼ��ʱ���ı�״����O2____________L��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(1)
(2)��A��ʹ��ˮ��ɫ�����ڴ�����������H2�ӳɵIJ�������к���4��������A���ܵĽṹ��ʽΪ(��дһ��) __________________________________��
(3)ij�л���ķ���ʽΪCxHyO2����x��ֵ��A�����е�̼ԭ�Ӹ�����ͬ����÷�����y�����ֵΪ__________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012��ɽ���ij�ݷ��ʵ����и߶�������ģ����Ի�ѧ�Ծ����������� ���ͣ������
��ÿ��2�֣���10�֣�ij��A 0.2 mol ����������ȫȼ�պ�����CO2��H2O��1.2 mol���Իش�
��1����A�ķ���ʽΪ_____________��
��2����ȡһ��������A��ȫȼ�պ�����CO2��H2O��3 mol������________g��A�μ��˷�Ӧ��ȼ��ʱ���ı�״���µ�����___________L��
��3������A����ʹ��ˮ��ɫ������һ��������������������ȡ����Ӧ����һ��ȡ����ֻ
��һ�֣�����A�Ľṹ��ʽΪ__________________��
��4������A��ʹ��ˮ��ɫ���ڴ��������£���H2�ӳɣ���ӳɲ��ᆳ�ⶨ�����к���
4��������A�����еĽṹ��ʽΪ______________��д��һ�ּ��ɣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�����ʡ�ױ��и�һ��ѧ�ڵڶ����¿���ѧ�Ծ��������棩 ���ͣ������
(12��)ij��A 0.2 mol�������г��ȼ�պ����ɻ�����B��C��1.2 mol���Իش�
(1)��A�ķ���ʽ��________________��
(2)��ȡһ��������A���ȼ�պ�����B��C��3 mol������________g��A�μ��˷�Ӧ��ȼ��ʱ���ı�״���µ�����________L��
(3)����A����ʹ��ˮ��ɫ������һ��������������������ȡ����Ӧ����һ�ȴ���ֻ��һ�֣�����A�Ľṹ��ʽ��________________��
��4������A��ʹ��ˮ��ɫ���ڴ����������������ӳɣ���ӳɲ�����ⶨ�����к���4����������A�Ľṹ��ʽ������__________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013���㽭ʡ�߶��ڶ����¿���ѧ�������Ծ� ���ͣ������
��14�֣�ij��A 0.2 mol ����������ȫȼ�պ�����CO2��H2O��1.2 mol���Իش�
��1����A�ķ���ʽΪ_____________��
��2����ȡһ��������A��ȫȼ�պ�����CO2��H2O��3 mol������________g��A�μ��˷�Ӧ��ȼ��ʱ���ı�״���µ�����___________L��
��3������A����ʹ��ˮ��ɫ������һ��������������������ȡ����Ӧ����һ��ȡ����ֻ��һ�֣�����A�Ľṹ��ʽΪ__________________��
��4������A��ʹ��ˮ��ɫ���ڴ��������£���H2�ӳɣ���ӳɲ��ᆳ�ⶨ�����к���4��������A�����еĽṹ��ʽΪ_______________________________________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com