
“Μ÷÷“‘ΜΤΆ≠ΩσΚΆΝρΜ«ΈΣ‘≠Νœ÷Τ»ΓΆ≠ΚΆΤδΥϊ≤ζΈοΒΡ–¬ΙΛ“’Θ§‘≠ΝœΒΡΉέΚœάϊ”Ο¬ ΫœΗΏΘ°Τδ÷ς“ΣΝς≥Χ»γœ¬ΘΚ
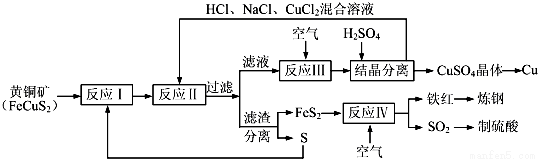
ΉΔΘΚΖ¥”ΠΔρΒΡάκΉ”ΖΫ≥Χ ΫΈΣCu2++CuS+4Cl-=2[CuCl2]-+S
«κΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®1Θ©Ζ¥”ΠΔώΒΡ≤ζΈοΈΣ____________Θ®ΧνΜ·―ß ΫΘ©Θ°
Θ®2Θ©Ζ¥”ΠΔσΒΡάκΉ”ΖΫ≥Χ ΫΈΣ____________ΘΜ
Θ®3Θ©“ΜΕ®Έ¬Ε»œ¬Θ§‘ΎΖ¥”ΠΔσΥυΒΟΒΡ»ή“Κ÷–Φ”»κœΓΝρΥαΘ§Ω…“‘Έω≥ωΝρΥαΆ≠ΨßΧεΘ§ΤδΩ…ΡήΒΡ‘≠“ρ «____________ΘΜ
Θ®4Θ©Ζ¥”ΠΔτ‘ΎΗΏΈ¬ΧθΦΰœ¬Ϋχ––Θ§Μ·―ßΖΫ≥Χ Ϋ «____________ΘΜ
Θ®5Θ©Ρ≥ΝρΥα≥ßΈΣ≤βΕ®Ζ¥”ΠΔτΥυΒΟΤχΧε÷–SO2ΒΡΧεΜΐΖ÷ ΐΘ§»Γ280mLΘ®“―’έΥψ≥…±ξΉΦΉ¥ΩωΘ©ΤχΧε―υΤΖ”κΉψΝΩFe2Θ®SO4Θ©3»ή“ΚΆξ»ΪΖ¥”ΠΚσΘ§”Ο≈®Ε»ΈΣ0.02000mol•L-1ΒΡK2Cr2O7±ξΉΦ»ή“ΚΒΈΕ®÷Ν÷’ΒψΘ§œϊΚΡK2Cr2O7»ή“Κ25.00mLΘ°“―÷ΣΘΚCr2O72-+Fe2++H+ΓζCr3++Fe3++H2OΘ®Έ¥≈δΤΫΘ©
ΔΌSO2Ά®»κFeΘ®SO4Θ©3»ή“Κ÷–Θ§ΖΔ…ζΖ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΈΣ____________ΘΜ
ΔΎΖ¥”ΠΔτΥυΒΟΤχΧε÷–SO2ΒΡΧεΜΐΖ÷ ΐΈΣ____________ΓΘ
 ‘ΡΕΝΩλ≥ΒœΒΝ–¥πΑΗ
‘ΡΕΝΩλ≥ΒœΒΝ–¥πΑΗ
| ΡξΦΕ | ΗΏ÷–ΩΈ≥Χ | ΡξΦΕ | ≥θ÷–ΩΈ≥Χ |
| ΗΏ“Μ | ΗΏ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ“Μ | ≥θ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏΕΰ | ΗΏΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θΕΰ | ≥θΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏ»ΐ | ΗΏ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ»ΐ | ≥θ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2015-2016―ßΡξΚ”±± ΓΦΫ÷ί –ΗΏ“Μœ¬ΤΎΡ©Μ·―ß ‘ΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚ―Γ‘ώΧβ
œ¬Ν–Ζ¥”Π÷–ΡήΦλ―ιΡρ“Κ÷–¥φ‘ΎΤœΧ―Χ«ΒΡ «Θ®Θ©
AΘ°Φ”Ϋπ τΡΤΘ§Ω¥ «Ζώ”–«βΤχΖ≈≥ω
BΘ°”κ–¬÷ΤΒΡCu(OH)2–ϋΉ«“ΚΜλΚœ÷σΖ–Θ§Ιέ≤λ”–ΈόΚλ…Ϊ≥ΝΒμ…ζ≥…
CΘ°”κ¥ΉΥαΚΆ≈®ΝρΥαΙ≤»»Θ§Ω¥ΡήΖώΖΔ…ζθΞΜ·Ζ¥”Π
DΘ°Φ”»κ±ΞΚΆΧΦΥαΡΤ»ή“Κ÷–Ω¥ «ΖώΖ÷≤ψ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2016ΫλΚΰ±± Γ…≥ –ΗΏ»ΐΗΏΩΦ«ΑΉνΚσ“ΜΨμάμΉέΜ·―ß ‘ΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚ―Γ‘ώΧβ
”ΟNaOH»ή“ΚΈϋ ’―ΧΤχ÷–ΒΡSO2Θ§ΫΪΥυΒΟΒΡNa2SO3»ή“ΚΫχ––ΒγΫβΘ§Ω…―≠ΜΖ‘Ό…ζNaOHΘ§Ά§ ±ΒΟΒΫH2SO4Θ§Τδ‘≠άμ»γΆΦΥυ ΨΘ®ΒγΦΪ≤ΡΝœΈΣ ·ΡΪΘ©ΓΘœ¬Ν–”–ΙΊ–π ω≤Μ’ΐ»ΖΒΡ «
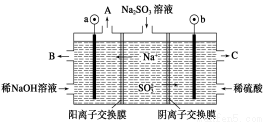
AΘ°ΆΦ÷–aΦΪΝ§Ϋ”Βγ‘¥ΒΡΗΚΦΪ
BΘ°AΩΎΖ≈≥ωΒΡΈο÷ ««βΤχΘ§CΩΎΖ≈≥ωΒΡΈο÷ «―θΤχ
CΘ°bΦΪΒγΦΪΖ¥”Π ΫΈΣΘΚSO32-Θ≠2eΘ≠ΘΪH2O===SO42-ΘΪ2HΘΪ
DΘ°ΒγΫβΙΐ≥Χ÷–“θΦΪ«χΦν–‘Οςœ‘‘ω«Ω
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2015-2016―ßΡξΥΡ¥® ΓΗΏ“Μœ¬ΒΎΕΰ¥ΈΕΈΩΦΜ·―ßΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚ―Γ‘ώΧβ
“―÷ΣΘΚΘ®1Θ©ZnΘ®sΘ©+1/2O2Θ®gΘ©= Z nOΘ®sΘ©Θ§ ΠΛH=-348.3kJ/mol
nOΘ®sΘ©Θ§ ΠΛH=-348.3kJ/mol
Θ®2Θ©2AgΘ®sΘ©+1/2 O2Θ®gΘ©= Ag2OΘ®sΘ©Θ§ ΠΛH=-31.0kJ/mol
‘ρZnΘ®sΘ©+ Ag2OΘ®sΘ©= ZnOΘ®sΘ©+ 2AgΘ®sΘ©ΒΡΠΛHΒ»”Ύ
2AgΘ®sΘ©ΒΡΠΛHΒ»”Ύ
A. -317.3kJ/mol B. -379.3kJ/mol C. -332.8 kJ/mol D. +317.3 kJ/mo l
l
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2016ΫλΫ≠Υ’ ΓΒ»»ΐ–ΘΗΏ»ΐΒΎΥΡ¥ΈΡΘΡβΜ·―ß ‘ΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚΧνΩ’Χβ
ΓΨΈο÷ ΫαΙΙ”κ–‘÷ ΓΩ¥ΩΆ≠‘ΎΙΛ“Β…œ÷ς“Σ”Οά¥÷Τ‘λΒΦœΏΓΔΒγΤς‘ΣΦΰΒ»Θ§Ά≠Ρή–Έ≥…Εύ÷÷-1ΚΆ+2ΦέΧ§ΒΡΜ·ΚœΈο«“ΤδΜ·ΚœΈο≥Θ¥χ”–―’…ΪΓΘ
Θ®1Θ©–¥≥ωΜυΧ§Cu+ΒΡΚΥΆβΒγΉ”≈≈≤Φ Ϋ____________ΘΜ
Θ®2Θ©»γΆΦΫαΙΙ «Ά≠ΒΡΡ≥―θΜ·ΈοΒΡΨßΧεΫαΙΙΒΡΉν–Γ÷ΊΗ¥ΒΞ‘ΣΘ§‘ρΗΟ―θΜ·ΈοΒΡΜ·―ß ΫΈΣ____________ΘΜ
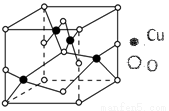
Θ®3Θ©œρΝρΥαΆ≠»ή“Κ÷–ΒΈΦ”Α±Υ°Μα…ζ≥…άΕ…Ϊ≥ΝΒμΘ§‘ΎΒΈΦ”Α±Υ°÷Ν≥ΝΒμΗ’ΚΟ»Ϊ≤Ω»ήΫβ ±Ω…ΒΟΒΫάΕ…Ϊ»ή“ΚΘ§ΦΧ–χœρΤδ÷–Φ”»κΦΪ–‘Ϋœ–ΓΒΡ““¥ΦΩ…“‘…ζ≥……νάΕ…ΪΒΡ[CuΘ®NH3Θ©4]SO4•H2O≥ΝΒμΓΘ
ΔΌΗΟ≥ΝΒμ÷–S‘≠Ή”ΒΡ‘”Μ·άύ–ΆΈΣ____________ΘΜ
ΔΎ–¥≥ω“Μ÷÷”κNH3Ζ÷Ή”ΜΞΈΣΒ»ΒγΉ”ΧεΒΡ―τάκΉ”ΒΡΜ·―ß Ϋ____________ΓΘ
Θ®4Θ©NH3ΒΡΖ–ΒψΗΏ”ΎPH3Θ§‘≠“ρ «____________
Θ®5Θ©CuOΒΡ»έΒψ±»CuClΒΡ»έΒψ____________Θ®ΧνΓΑΗΏΓ±ΜρΓΑΒΆΓ±Θ©Θ§Τδ‘≠“ρ «____________ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2016ΫλΫ≠Υ’ ΓΒ»»ΐ–ΘΗΏ»ΐΒΎΥΡ¥ΈΡΘΡβΜ·―ß ‘ΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚ―Γ‘ώΧβ
œ¬Ν–”–ΙΊΥΒΖ®’ΐ»ΖΒΡ «
AΘ°Ρ≥Έ¬Ε»œ¬Θ§1L pH=8ΒΡ¥ΩΥ°÷–Κ§OH-ΈΣ10 -6rnol
BΘ°ΒγΫβΨΪΝΕΆ≠Ιΐ≥Χ÷–Θ§―τΦΪ÷ ΝΩΒΡΦθ…Ό÷Β”κ“θΦΪ÷ ΝΩΒΡ‘ωΦ”÷Β“ΜΕ®œύΒ»
CΘ°Κœ≥…Α±ΒΡΖ¥”Π «Ζ≈»»Ζ¥”ΠΘ§‘ρ≤…”ΟΒΆΈ¬ΧθΦΰΩ…“‘ΧαΗΏΑ±ΒΡ…ζ≥…ΥΌ¬
DΘ°CH3COOH»ή“ΚΦ”Υ°œΓ ΆΚσΘ§»ή“Κ÷–cΘ®CH3COOHΘ©/cΘ®OH-Θ©ΒΡ÷ΒΦθ–Γ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2016ΫλΫ≠Υ’ ΓΒ»»ΐ–ΘΗΏ»ΐΒΎΥΡ¥ΈΡΘΡβΜ·―ß ‘ΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚ―Γ‘ώΧβ
œ¬Ν–”–ΙΊΈο÷ ΒΡ–‘÷ Μρ”Π”ΟΨυ’ΐ»ΖΒΡ «
AΘ°Εΰ―θΜ·ΙηΈΣΥα–‘―θΜ·ΈοΘ§Ω…”Ο”Ύ÷ΤΉωΦΤΥψΜζ–ΨΤ§
BΘ°Ά≠ΒΡΫπ τΜνΤΟ–‘±»Χζ≤νΘ§Ω…‘ΎΚΘ¬÷ΆβΩ«…œΉΑ»τΗ…Ά≠Ωι“‘ΦθΜΚΤδΗ· ¥
CΘ°ZnΨΏ”–ΜΙ‘≠–‘ΚΆΒΦΒγ–‘Θ§Ω…”ΟΉς–ΩΟΧΗ…Βγ≥ΊΒΡΗΚΦΪ≤ΡΝœ
DΘ°≈®ΝρΥαΨΏ”–Ά―Υ°–‘Θ§Ω…”Ο”ΎΗ…‘οNH3ΓΔSO2Β»
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2016ΫλΚΰ±± ΓΗΏ»ΐ5‘¬ΡΘΡβ»ΐάμΉέΜ·―ß ‘ΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚΧνΩ’Χβ
ν―ΓΔΗθΓΔΧζΓΔΡχΓΔΆ≠Β»Ϋπ τΦΑΤδΜ·ΚœΈο‘ΎΙΛ“Β…œ”–÷Ί“Σ”ΟΆΨΓΘ
Θ®1Θ©ν―ΧζΚœΫπ «ν―œΒ¥Δ«βΚœΫπΒΡ¥ζ±μΘ§ΗΟΚœΫπΨΏ”–Ζ≈«βΈ¬Ε»ΒΆΓΔΦέΗώ ÷–Β»”≈ΒψΓΘ
ΔΌTiΒΡΜυΧ§‘≠Ή”ΦέΒγΉ”≈≈≤Φ ΫΈΣ________________ΓΘ
ΔΎFeΒΡΜυΧ§‘≠Ή”Ι≤”–________÷÷≤ΜΆ§ΡήΦΕΒΡΒγΉ”ΓΘ
Θ®2Θ©÷Τ±ΗCrO2Cl2ΒΡΖ¥”ΠΈΣK2Cr2O7ΘΪ3CCl4===2KClΘΪ2CrO2Cl2ΘΪ3COCl2ΓϋΓΘ
ΔΌ…œ ωΜ·―ßΖΫ≥Χ Ϋ÷–Ζ«Ϋπ τ‘ΣΥΊΒγΗΚ–‘”…¥σΒΫ–ΓΒΡΥ≥–ρ «__________(”Ο‘ΣΥΊΖϊΚ≈±μ Ψ)ΓΘ
ΔΎCOCl2Ζ÷Ή”÷–Υυ”–‘≠Ή”Ψυ¬ζΉψ8ΒγΉ”ΙΙ–ΆΘ§COCl2Ζ÷Ή”÷–Π“ΦϋΚΆΠ–ΦϋΒΡΗω ΐ±»ΈΣ_____Θ§÷––Ρ‘≠Ή”ΒΡ‘”Μ·ΖΫ ΫΈΣ________ΓΘ
Θ®3Θ©NiOΓΔFeOΒΡΨßΧεΫαΙΙΨυ”ꬻ̷ΡΤΒΡΨßΧεΫαΙΙœύΆ§Θ§Τδ÷–Ni2ΘΪΚΆFe2ΘΪΒΡάκΉ”ΑκΨΕΖ÷±πΈΣ690pmΚΆ780pmΓΘ‘ρ»έΒψΘΚNiO________(ΧνΓΑ>Γ±ΓΔΓΑ<Γ±ΜρΓΑΘΫΓ±)FeOΓΘ
Θ®4Θ©NiΚΆLaΒΡΚœΫπ «ΡΩ«Α Ι”ΟΙψΖΚΒΡ¥Δ«β≤ΡΝœΘ§ΨΏ”–¥σ»ίΝΩΓΔΗΏ ΌΟϋΓΔΡΆΒΆΈ¬Β»ΧΊΒψΘ§‘Ύ»’±ΨΚΆ÷–Ιζ“― Βœ÷ΝΥ≤ζ“ΒΜ·ΓΘΗΟΚœΫπΒΡΨßΑϊΫαΙΙ»γΆΦΥυ ΨΓΘ
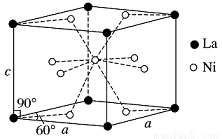
ΔΌΗΟΨßΧεΒΡΜ·―ß ΫΈΣ________________ΓΘ
ΔΎ“―÷ΣΗΟΨßΑϊΒΡΡΠΕϊ÷ ΝΩΈΣM gΓΛmolΘ≠1Θ§ΟήΕ»ΈΣd gΓΛcmΘ≠3ΓΘ…ηNAΈΣΑΔΖϋΦ”Β¬¬ό≥Θ ΐΒΡ÷ΒΘ§‘ρΗΟΨßΑϊΒΡΧεΜΐ «________ cm3(”ΟΚ§MΓΔdΓΔNAΒΡ¥ζ ΐ Ϋ±μ Ψ)ΓΘ
ΔέΗΟΨßΧεΒΡΡΎ≤ΩΨΏ”–Ω’œΕΘ§«“ΟΩΗωΨßΑϊΒΡΩ’œΕ÷–¥Δ¥φ6Ηω«β‘≠Ή”±»ΫœΈ»Ε®ΓΘ“―÷ΣΘΚaΘΫ511 pmΘ§cΘΫ397 pmΘΜ±ξΉΦΉ¥Ωωœ¬«βΤχΒΡΟήΕ»ΈΣ8.98ΓΝ10Θ≠5 gΓΛcmΘ≠3ΘΜ¥Δ«βΡήΝΠΘΫ ΓΘ»τΚω¬‘Έϋ«β«ΑΚσΨßΑϊΒΡΧεΜΐ±δΜ·Θ§‘ρΗΟ¥Δ«β≤ΡΝœΒΡ¥Δ«βΡήΝΠΈΣ_______ΓΘ
ΓΘ»τΚω¬‘Έϋ«β«ΑΚσΨßΑϊΒΡΧεΜΐ±δΜ·Θ§‘ρΗΟ¥Δ«β≤ΡΝœΒΡ¥Δ«βΡήΝΠΈΣ_______ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2016ΫλΝ…Ρΰ ΓΗΏ»ΐ»Ϊ’φΡΘΡβάμΉέΜ·―ß ‘ΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚΧνΩ’Χβ
ΦΉ¥Φ «÷Ί“ΣΒΡΜ·ΙΛ‘≠ΝœΘ§”÷Ω…≥ΤΈΣ»ΦΝœΓΘΙΛ“Β…œάϊ”ΟΚœ≥…Τχ(÷ς“Σ≥…Ζ÷ΈΣCOΓΔCO2ΚΆH2)‘Ύ¥ΏΜ·ΦΝΒΡΉς”Οœ¬Κœ≥…ΦΉ¥ΦΘ§ΖΔ…ζΒΡ÷ς“ΣΖ¥”Π»γœ¬ΘΚ
ΔΌCO(g)+2H2(g) CH3OH(g) ΓςH
CH3OH(g) ΓςH
ΔΎCO2(g)+3H2(g) CH3OH(g)+H2O(g) ΓςH2=-58kJ•mol-1
CH3OH(g)+H2O(g) ΓςH2=-58kJ•mol-1
ΔέCO2(g)+H2(g) CO(g)+H2O(g) ΓςH3=+41kJ•mol-1
CO(g)+H2O(g) ΓςH3=+41kJ•mol-1
ΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®1Θ©“―÷ΣΖ¥”ΠΔΌ÷–ΒΡœύΙΊΒΡΜ·―ßΦϋΦϋΡή ΐΨί»γœ¬ΘΚ

‘ρx=____________ΘΜ
Θ®2Θ©»τTΓφ ±ΫΪ6molCO2ΚΆ8molH2≥δ»κ2LΟή±’»ίΤς÷–ΖΔ…ζΖ¥”ΠΔΎΘ§≤βΒΟH2ΒΡΈο÷ ΒΡΝΩΥφ ±Φδ±δΜ·ΙΊœΒ»γΆΦ÷–Ή¥Χ§I(ΆΦ÷– ΒœΏ)Υυ ΨΓΘΆΦ÷– ΐΨίA(1Θ§6)¥ζ±μ‘Ύ1min ±H2ΒΡΈο÷ ΒΡΝΩ «6molΓΘ
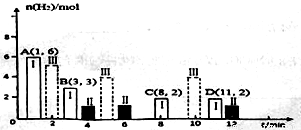
ΔΌTΓφ ±Ή¥Χ§IΧθΦΰœ¬Θ§0~3minΡΎCH3OHΒΡΤΫΨυΖ¥”ΠΥΌ¬ v=_______mol/(LΓΛmin)Θ§ΤΫΚβ≥Θ ΐK=_____ΘΜ
ΔΎΤδΥϊΧθΦΰ≤Μ±δ ±Θ§ΫωΗΡ±δΡ≥“ΜΧθΦΰΚσ≤βΒΟH2ΒΡΈο÷ ΒΡΝΩΥφ ±Φδ±δΜ·»γΆΦ÷–Ή¥Χ§ΔρΥυ ΨΘ§‘ρΗΡ±δΒΡΧθΦΰΩ…Ρή «_____ΘΜ
ΔέΤδΥϊΧθΦΰ≤Μ±δΘ§ΫωΗΡ±δΈ¬Ε» ±Θ§≤βΒΟH2ΒΡΈο÷ ΒΡΝΩΥφ ±Φδ±δΜ·»γΆΦ÷–Ή¥Χ§ΔσΥυ ΨΘ§‘ρΉ¥Χ§ΔσΕ‘”ΠΒΡΈ¬Ε»_______(ΧνΓΑΘΨΓ±ΓΔΓΑΘΦΓ±ΜρΓΑ=Γ±)TΓφΘΜ
Δή»τΉ¥Χ§ΔρΒΡΤΫΚβ≥Θ ΐΈΣK2Θ§Ή¥Χ§ΔσΒΡΤΫΚβ≥Θ ΐΈΣK3Θ§‘ρK2_______(ΧνΓΑΘΨΓ±ΓΔΓΑΘΦΓ±ΜρΓΑ=Γ±)K3ΘΜ
Δί“ΜΕ®Έ¬Ε»œ¬Ά§Θ§¥ΥΖ¥”Π‘ΎΚψ»ί»ίΤς÷–Ϋχ––Θ§Ρή≈–ΕœΗΟΖ¥”Π¥οΒΫΜ·―ßΤΫΚβ“άΨίΒΡ «_______ΓΘ
aΘ°»ίΤς÷–―Ι«Ω≤Μ±δ
bΘ°ΦΉ¥ΦΚΆΥ°’τΤϊΒΡΧεΜΐ±»±Θ≥÷≤Μ±δ
cΘ°v’ΐ(H2)=3vΡφCH3OH)
dΘ°2ΗωC=OΕœΝ―ΒΡΆ§ ±”–6ΗωHΓΣHΕœΝ―
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΙζΦ ―ß–Θ”≈―Γ - ΝΖœΑ≤αΝ–±μ - ‘ΧβΝ–±μ
Κΰ±± ΓΜΞΝΣΆχΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΤΫΧ® | Άχ…œ”–ΚΠ–≈œΔΨΌ±®Ή®«χ | Βγ–≈’©Τ≠ΨΌ±®Ή®«χ | …φάζ Ζ–ιΈό÷ς“ε”–ΚΠ–≈œΔΨΌ±®Ή®«χ | …φΤσ«÷»®ΨΌ±®Ή®«χ
ΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΒγΜΑΘΚ027-86699610 ΨΌ±®” œδΘΚ58377363@163.com