
��7�֣�ÿ��1�֣�A��B��C��DΪͬһ���ڵ�����Ԫ�أ�ԭ����������������֪0.2ĦA�����ַ�Ӧʱ���ڱ���¿�����2.24��������B��������ȿ�����ǿ���ֿ�����ǿ����Һ��C��D�����ӵĵ��Ӳ�ṹ���ԭ����ͬ��C����̬�⻯����C�ĵͼ������ﷴӦ���ֿɵõ�C�ĵ��ʡ��Իش�
��1��A��B��C��D��Ԫ�ط��ŷֱ�Ϊ______��____ �� ��______��
��2���õ���ʽ��ʾA��C�γɻ�����Ĺ��̣�
_________________________________________________��
��3��д��B����������A���������ﷴӦ�����ӷ���ʽ��
_________________________________ ��
��4��д��ʵ�����Ʊ�D�ĵ��ʵĻ�ѧ��Ӧ����ʽ��
__________________________________________��
 ������������Ӧ����ϵ�д�
������������Ӧ����ϵ�д� ͬ����չ�Ķ�ϵ�д�
ͬ����չ�Ķ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2010��2011������ʡ�������и�һ��ѧ��6���¿���ѧ�Ծ� ���ͣ������
��7�֣�ÿ��1�֣�A��B��C��DΪͬһ���ڵ�����Ԫ�أ�ԭ����������������֪0.2ĦA�����ַ�Ӧʱ���ڱ���¿�����2.24��������B��������ȿ�����ǿ���ֿ�����ǿ����Һ��C��D�����ӵĵ��Ӳ�ṹ���ԭ����ͬ��C����̬�⻯����C�ĵͼ������ﷴӦ���ֿɵõ�C�ĵ��ʡ��Իش�
��1��A��B��C��D��Ԫ�ط��ŷֱ�Ϊ______��____ �� ��______��
��2���õ���ʽ��ʾA��C�γɻ�����Ĺ��̣�
_________________________________________________��
��3��д��B����������A���������ﷴӦ�����ӷ���ʽ��
_________________________________ ��
��4��д��ʵ�����Ʊ�D�ĵ��ʵĻ�ѧ��Ӧ����ʽ��
__________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ���Ĵ�ʡ�Ű���ѧ��һ��ѧ�����п��Ի�ѧ�Ծ����������� ���ͣ������
��7�֣�ÿ��1�֣���ͼ��ʾ����֪AԪ�ص���ͻ��ϼ�Ϊ��3�ۣ�������������ﺬ��56.21%��ԭ�Ӻ�������������������1��������ӦԪ�ط��Ż�ѧʽ�ش�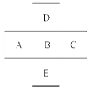
(1)д�����ǵ�Ԫ�ط��ţ�
A__________��B__________��C___________��D__________��
(2)A��B��C��ԭ�Ӱ뾶��С�����˳����________________________________��
(3)A��B��C������������Ӧˮ�����������ǿ������˳���� ��D��B��Ԫ�ص���̬�⻯���ȶ��Դ�С˳���� .
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ����������ʦר�ã�2010�꽭��ʡ�ϸ߶��и߶���ѧ�����п��Ի�ѧ���� ���ͣ������
��7�֣�ÿ��1�֣������廯����A��B��Ϊͬ���칹�壬B�Ľṹ��ʽ��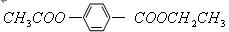 ��A���٢ڷ�Ӧ��C��D��E��B���٢ڷ�
��A���٢ڷ�Ӧ��C��D��E��B���٢ڷ�
Ӧ��E��F��H��������Ӧ���� ���������ʼ����ϵ��ͼ��
���������ʼ����ϵ��ͼ��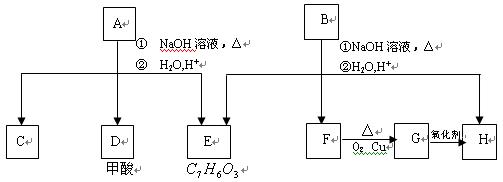
��1��E�к��й����ŵ�������__________________��
��2��A�����ֿ��ܵĽṹ����ṹ��ʽ�ֱ�Ϊ_____________��______________��
��3��C��D�� F��G��H�������л�Ϊͬϵ�����___________��__________��
F��G��H�������л�Ϊͬϵ�����___________��__________��
��4��C��Ũ
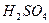 �����¼��ȷ�����Ӧ�������ﲻʹ��ˮ��ɫ���������ʵĽṹ��ʽΪ��______________________________��
�����¼��ȷ�����Ӧ�������ﲻʹ��ˮ��ɫ���������ʵĽṹ��ʽΪ��______________________________��
��5��д����G����������F�ķ�Ӧ����_________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010-2011������ʡ��һ��ѧ��6���¿���ѧ�Ծ� ���ͣ������
��7�֣�ÿ��1�֣�A��B��C��DΪͬһ���ڵ�����Ԫ�أ�ԭ����������������֪0.2ĦA�����ַ�Ӧʱ���ڱ���¿�����2.24��������B��������ȿ�����ǿ���ֿ�����ǿ����Һ��C��D�����ӵĵ��Ӳ�ṹ���ԭ����ͬ��C����̬�⻯����C�ĵͼ������ﷴӦ���ֿɵõ�C�ĵ��ʡ��Իش�
��1��A��B��C��D��Ԫ�ط��ŷֱ�Ϊ______��____ �� ��______��
��2���õ���ʽ��ʾA��C�γɻ�����Ĺ��̣�
_________________________________________________��
��3��д��B����������A���������ﷴӦ�����ӷ���ʽ��
_________________________________ ��
��4��д��ʵ�����Ʊ�D�ĵ��ʵĻ�ѧ��Ӧ����ʽ��
__________________________________________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com