
�±��Dz�ͬ�¶���ˮ�����ӻ�������
|
�¶�/�� |
25 |
t1 |
t2 |
|
ˮ�����ӻ����� |
1��10��14 |
KW |
1��10��12 |
�Իش��������⣺
��1����25��<t1<t2����KW__________1��10��14���>������<�����������жϵ�������
��
��2��25�� �£���pH��13������������Һ��pH��1��ϡ����������ϣ����û����Һ��pH��__________��
��3��������������ʵ���Ũ�ȵĴ��������������Һ��Ϻ���Һ�� ������ԡ��������ԡ����ԡ�������Һ��c(Na+) c(CH3COO-)�����������������������
��4��pH��3�Ĵ����pH��11������������Һ�������Ϻ���Һ�� �ԣ���ᡱ�� ���С��������Һ��c(Na+) c(CH3COO-)[�����������������] ��
(1) >�� ��Ϊˮ�ĵ��������ȷ�Ӧ���¶����ߣ�ˮ�ĵ���̶��������ӻ�Ҳ����
(2) 7 (3) ���� > ��4������ ��
��������
�����������1��ˮ�ĵ��������һ�����ȹ��̣���ˣ������¶ȣ��ٽ����룬��Ӧ��ˮ�����ӻ�����Ҳ��֮����
��2��25����ˮ�����ӻ�����Ϊ1��10��14����pH��13������������Һ��pH��1��ϡ����������ϣ����û����Һ��pHΪ7.
��3��������������ʵ���Ũ�ȵĴ��������������Һ��Ϻ�������Һ������ΪCH3COONa��Ϊǿ�������Σ����Ӧ����Һ�Լ��ԡ�����CH3COO-��ˮ�⣬���c(Na+)��c(CH3COO-)��
��4��pH��3�Ĵ����pH��11������������Һ�������Ϻ�������Һ����ΪCH3COOH��CH3COONa����c(CH3COOH)��c(CH3COONa)����Һ�����ԣ�������Һ��CH3COOH�ĵ��룬���c(Na+)��c(CH3COO-)��
���㣺�������Һ��ˮ�ĵ���
����������������Ķ��ǻ����⣬�������տα��Ļ������ݼ��ɺܺõĽ�𡣴�����Ӧע��һ�����⣺��pH��3�Ĵ��ᡱ�����ڴ��������ᣬ���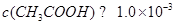 ��
��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �¶�/�� | 25 | t1 | t2 |
| ˮ�����ӻ� | 1��10-14 | a | 1��10-12 |
| SO | 2- 4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �¶ȣ��棩 | 25 | t1 | t2 |
| ˮ�����ӻ����� | 1��10-14 | �� | 1��10-13 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �¶�/�� | 25 | t1 | t2 |
| Kw/mol2?L-2 | 1��10-14 | a | 1��10-12 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �¶�/�� | 25 | t1 | t2 |
| ˮ�����ӻ����� | 1��10-14 | KW | 1��10-12 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �¶�/�� | 25 | t1 | t2 |
| ˮ�����ӻ����� | 1��10-14 | KW | 1��10-12 |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com