
���ݱ�����Ϣ�ش��������⡣
| Ԫ�� | Si | P | S | Cl |
| ���������� ��Ӧ������ | ���� | �������������ܷ�Ӧ | ���� | ���ջ��ȼʱ������ը������ |
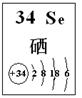
| ��� | �����Ʋ� | ��ѧ����ʽ |
| 1 | ������ | H2SeO3��4HI=Se����2I2��3H2O |
| 2 | | |
| 3 | | |
��1����3����VIA��2���˵������������������
��3��P4��s��+6H2(g)=4PH3(g) ��H=+37.2 kJ��mol-1
��4����d
��1 ����Ԥ�� ��ѧ����ʽ 2 ��ԭ�� H2SeO3+Cl2+H2O=" " H2SeO4+2HCl 3 ���� H2SeO3+2NaOH=Na2 SeO3+2H2O.
���������������1������2���μ��𰸣���3����д����̬���ף�P4����H2��Ӧ������̬�⻯��Ļ�ѧ����ʽ��P4+6H2=4PH3��ͨ����ѧ����ʽ���Կ�����1mol P4��H2��Ӧ����4 mol PH3��������������������Ӧ����4 mol PH3ʱ����Ӧ�ų�������Ϊ��4��+9.3 kJ��mol-1=+37.2 kJ��mol-1 �������Ȼ�ѧ����ʽΪ��
P4��s��+6H2(g)=4PH3(g) ��H=+37.2 kJ��mol-1��4����a.����ͬ���ں�ͬ����Ԫ��ԭ�Ӱ뾶�ĵݱ���ɣ���֪��ȷ��ϵӦΪ��Se��P��S������b.ͬ����Ԫ����̬�⻯����ȶ������϶������μ���,��ȷ��ӦΪ: H2Se��H2S ������c.Ӧ��Ϊ����������Ӧ��ˮ��������ԣ�����H2SeO4��HClO4,��ǽ�����Se��Cl������d.����ͬ����Ԫ�ص����ʵ������ԣ����ѵ�֪d��ȷ��
��4���о�H2SeO3�Ļ�ѧ���ʿɴ�H2SO3�Ļ�ѧ���ʽ������ƣ��������л�ԭ�ԣ���������������Ӧ���ɴ˿��ƶ�H2SeO3Ҳ�л�ԭ�ԣ���ѧ����Ϊ�� H2SeO3+Cl2+H2O= H2SeO4+2HCl�������������ԣ���������Ӧ���������H2SeO3Ҳ�ɴ����ʣ���ѧ����ʽΪ��H2SeO3+2NaOH=Na2 SeO3+2H2O.
���㣺����Ԫ�����ڱ���Ԫ�������ɡ�ԭ�ӽṹ��ԭ�Ӱ뾶�Ĵ�С�Ƚϡ��Ȼ�ѧ���̵���д



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ڱ���ǰ36��Ԫ��A��B��C��D��E��F��G��H�����ǵ�ԭ������������������B��C��DΪͬһ���ڣ�E��F��GΪͬһ���ڣ�A��E��D��G�ֱ�Ϊͬһ���壻A��B��ɵĻ�������һ���������壻DԪ��ԭ�ӵ������������Ǵ�����������3����E����������ԭ�Ӱ뾶����Ԫ�أ���ϡ�������⣩��FԪ�ص����Ӱ뾶��ͬ����Ԫ���γɵļ���������С�ģ�HԪ�ػ�̬ԭ�ӵ�M��ȫ������N��û�гɶԵ��ӣ�ֻ��һ��δ�ɶԵ��ӡ�
��1��E2D2�����л�ѧ����������__________________
��2��һ�������£�A2������C2�����ַ�Ӧ������6.8 g�����壬�ų�18.44 kJ��������÷�Ӧ���Ȼ�ѧ����ʽΪ��_________________________________________����������ӵĿռ乹��Ϊ_________________������C���ӻ���ʽΪ_______________��
��3��C��E��ɵ�һ�����ӻ��������ˮ��Ӧ�������ּ�÷�Ӧ�Ļ�ѧ����ʽ��
��
��4��H�Ļ�̬ԭ�ӵĵ����Ų�ʽΪ__________________________��֪H2+���루2���е��������ˮ��Һ�γ�������д����������������ӵĽṹ��ʽ��_________________���ڴ�������ˮ��Һ�м����Ҵ���������_______________________________________��
��5����������GD2ͨ��EDA��Һ��������Һ������Ũ�ȵĴ�С˳��Ϊ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ�Dz��ֶ�����Ԫ�صij������ϼ���ԭ�������Ĺ�ϵͼ��
��1��Ԫ��A�����ڱ��е�λ�� ��
��2���õ���ʽ��ʾD2G���γɹ��� ����������ѧ������Ϊ ��
��3��C2����D+��G2�����Ӱ뾶�ɴ�С˳���� �������ӷ��Żش𣩡�
��4��ijͬѧ���ʵ��֤��A��B��F�ķǽ�����ǿ����ϵ��
����Һa��b�ֱ�Ϊ �� ��
����Һc�е����ӷ���ʽΪ ��
�����ԭ�ӽṹ�ĽǶȽ��ͷǽ�����B��A��ԭ�� ��
��5����0.5 mol D2C2Ͷ��100 mL 3 mol/L ECl3��Һ�У�
��ת�Ƶ��ӵ����ʵ���Ϊ ��
����һ�����ӷ���ʽ��ʾ�÷�Ӧ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
U��V��W��X��Y��Z��ԭ������������������ֳ���Ԫ�ء�Y�ĵ�����W2��ȼ�յIJ����ʹƷ����Һ��ɫ��Z��WԪ���γɵĻ�����Z3W4���д��ԡ�U�ĵ�����W2��ȼ�տ�����UW��UW2�������塣X�ĵ�����һ�ֽ������ý�����UW2�о���ȼ�����ɺڡ������ֹ��塣
��ش��������⣺
��1��V�ĵ��ʷ��ӵĽṹʽΪ ��XW�ĵ���ʽΪ ��
��2��ZԪ�������ڱ��е�λ���� ��
��3��U��V��W�γɵ�10�����⻯���У�U��V���⻯��е�ϵ͵��ǣ�д��ѧʽ�� ��
��4��д��X�ĵ�����UW2��ȼ�յĻ�ѧ����ʽ������ʾ���ӵ�ת�Ʒ������ĿΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪��X��Y��Z��R���ֶ�����Ԫ��ԭ��������������X����̬�⻯��ˮ��Һ�ʼ��ԣ�Yԭ�ӵ�L���������K���������3����Z��R�ĵ�����һ����������ͭ��Ӧ����Cu2Z��CuR2�������Ҫ����ա�
��1������X��ԭ�ӽṹʾ��ͼ ��R����������Ӧˮ����Ļ�ѧʽΪ ��
��2��Y��Z��R����Ԫ����ԭ�Ӱ뾶������ ����Ԫ�ط��ű�ʾ����
��3��X��̬�⻯����Z������������Ӧˮ���ﷴӦ���ɵIJ�����������ѧ��������Ϊ ������ò����������ӵ�ʵ�鷽���� ��
��4��R�ĵ���ͨ�뵽����ʯ��ˮ�з�Ӧ�����ӷ���ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��A��B��C��D��E��F���ֶ�����Ԫ�أ���Ԫ��������Ϣ���±���
| Ԫ�ر�� | Ԫ��������Ϣ |
| A | A�ĵ������ܶ���С������ |
| B | B�ĵ���������ˮ���ҷ�Ӧ������ǿ������Һ�к������ֵ�������ͬ������������ |
| C | C��ԭ�����������������ڲ������������ |
| D | D��Bͬ���ڣ���������D�ļ����Ӱ뾶��С |
| E | B��C��E��ɵ�36���ӵĻ�����Y�Ǽ�������������Ҫ�ɷ� |
| F | FԪ���������������۵Ĵ�����Ϊ4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�±�ΪԪ�����ڱ���һ���֣������Ԫ�آ١����ڱ��е�λ�ã�����Ӧ��ѧ����ش��������⣺
| ������ | IA | | 0 | |||||
| 1 | �� | ��A | ��A | ��A | ��A | ��A | ��A | |
| 2 | | | | �� | �� | �� | | |
| 3 | �� | | �� | �� | | �� | �� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��1���ǽ�������A��ʵ�����ﳣ�õĶ��Ե缫���ϣ�AԪ��ԭ�ӻ�̬ʱ�ĵ����Ų�ͼΪ ��
��2��Bԭ�ӻ�̬ʱ2pԭ�ӹ������3��δ�ɶԵ��ӣ�����������ȷ���� ������ĸ��ţ�
a��B����̬�⻯��ķе���ͬ��Ԫ�ص��⻯�������
b��B�ĵ�һ�����ܡ��縺�Զ���A�Ĵ�
c��A����̬�⻯���B����̬�⻯���ȶ�
d��A�ĺ�����һ����B�ĺ���������ǿ
��3��ͭ���ʼ��仯�����ںܶ���������Ҫ����;���糬ϸͭ�ۿ�Ӧ���ڵ�����ϡ������������С���ϸͭ�۵�ij�Ʊ��������£�
��[Cu(NH3)4]SO4�У�N��O��S����Ԫ�صĵ�һ�����ܴ�С�����˳��Ϊ�� ��
��SO42���ռ乹���� �� д��һ����SO32����Ϊ�ȵ�����ķ��� ��
��NH4CuSO3�еĽ��������ӵĽṹʾ��ͼΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
X��R��Ԫ�����ڱ��еĶ�����Ԫ�أ������ʻ�ṹ��Ϣ���±���
| Ԫ�� | X | Y | Z | W | R |
| ������Ϣ | ���γ�+7�۵Ļ����� | �ճ������г����������ۻ�ʱ��������,������һ��Ĥ���� | ͨ��״�������γɶ����������ȶ���˫ԭ�ӷ��� | ��ɫ��ӦΪ��ɫ | λ�ڵ�IVA�����γɻ�������������Ԫ�� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com