


 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�����ʡ�����ص���ѧ��һ�^����������ѧ�Ծ����������� ���ͣ�ʵ����
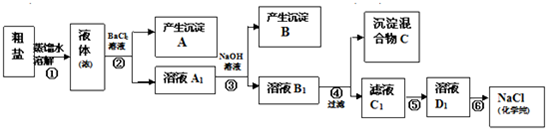
��12�֣�ijУ̽��ѧϰС��ͬѧ�ú�����������(��ҪΪ������ɳ��CaCl2��MgCl2��Na2SO4��)�Ĵ�����ȡ����ѧ��������NaCl��ʵ��ǰ������������·���(��ͼ)��
(1)��д�������ڢܡ��ݲ������Լ����Ƽ��ڢ��������ƣ��� ���� ���� ��
(2) ���������C�Ļ�ѧ�ɷ���(�����ֺͻ�ѧʽ��ʾ)�� ��
(3)д���ڢݲ������п��ܷ�����Ӧ�����ӷ���ʽ��
��
(4)��������������ڢݲ�ʵ���Ƿ�ﵽ��Ŀ�ģ�
��
(5)����Ϊ���������Щ���������Ӱ��ʵ������ ��
(6)��ͬѧ��Ϊ����ʵ����Ʋ�����Լ����������һ�����룺
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015��ɽ��ʡ�ij��и�һ��ѧ����У������ĩ��⻯ѧ�Ծ��������棩 ���ͣ�ʵ����
ijУ̽��ѧϰС��ͬѧ�ú�����������(��ҪΪ������ɳ��CaCl2��MgCl2��Na2SO4��)�Ĵ�����ȡ����ѧ��������NaCl��ʵ��ǰ������������·���(��ͼ)��
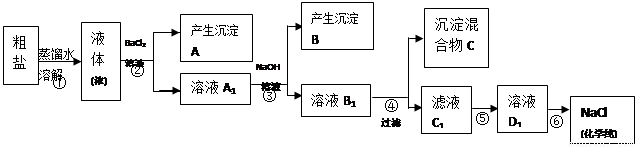
(1)��д�������ڢܡ��ݲ������Լ����Ƽ��ڢ��������ƣ��� ���� ���� ��
(2) ���������C�Ļ�ѧ�ɷ���(�����ֺͻ�ѧʽ��ʾ)�� ��
(3)д���ڢݲ������п��ܷ�����Ӧ�����ӷ���ʽ��
��
(4)��������������ڢݲ�ʵ���Ƿ�ﵽ��Ŀ�ģ�
��
(5)����Ϊ���������Щ���������Ӱ��ʵ������ ��
(6)��ͬѧ��Ϊ����ʵ����Ʋ�����Լ����������һ�����룺
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015�����ʡ��һ�^����������ѧ�Ծ��������棩 ���ͣ�ʵ����
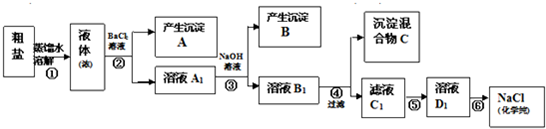
��12�֣�ijУ̽��ѧϰС��ͬѧ�ú�����������(��ҪΪ������ɳ��CaCl2��MgCl2��Na2SO4��)�Ĵ�����ȡ����ѧ��������NaCl��ʵ��ǰ������������·���(��ͼ)��

(1)��д�������ڢܡ��ݲ������Լ����Ƽ��ڢ��������ƣ��� ���� ���� ��
(2) ���������C�Ļ�ѧ�ɷ���(�����ֺͻ�ѧʽ��ʾ)�� ��
(3)д���ڢݲ������п��ܷ�����Ӧ�����ӷ���ʽ��
��
(4)��������������ڢݲ�ʵ���Ƿ�ﵽ��Ŀ�ģ�
��
(5)����Ϊ���������Щ���������Ӱ��ʵ������ ��
(6)��ͬѧ��Ϊ����ʵ����Ʋ�����Լ����������һ�����룺
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ��ʴ���
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com