
| A���٢ۢ� | B���ڢ� | C���ڢ� | D���ܢݢ� |
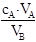 (VB��ȷ��ȡ�Ĵ���Һ�����,cA������Һ��Ũ��)��VAƫ��cBƫ��;��VAƫС��cBƫ�ͣ����÷�̪��ָʾ������Ӱ�죻�ڵζ�����ˮϴ��δ�ñ���Һ��ϴ��װ�����Һ����Һ��ϡ�ͣ��������ƫ����ҹŨ��ƫ�ߣ�����ƿʢ����Һǰ��δ�ô���Һ��ϴ����Ӱ�죻�ܱ�Һ����ʱ���ζ�ǰ���ӣ��ζ����ӣ���Һ���ƫС������ҹŨ��ƫ�ͣ�������ȡ����Һ�ĵζ���δ�ô���Һ��ϴ������ҹ��ϡ�ͣ�Ũ��ƫ�ͣ�����ҡ����ƿ�Ĺ����в���������һС������Һ�����ñ�Һ�����ƫС������ҹŨ��ƫ�ͣ��ߵζ����յ�ʱ���ֵζ��ܼ��촦����һ��Һ�Σ���Һ���ƫ����ҹŨ��ƫ�ߣ�ѡC��
(VB��ȷ��ȡ�Ĵ���Һ�����,cA������Һ��Ũ��)��VAƫ��cBƫ��;��VAƫС��cBƫ�ͣ����÷�̪��ָʾ������Ӱ�죻�ڵζ�����ˮϴ��δ�ñ���Һ��ϴ��װ�����Һ����Һ��ϡ�ͣ��������ƫ����ҹŨ��ƫ�ߣ�����ƿʢ����Һǰ��δ�ô���Һ��ϴ����Ӱ�죻�ܱ�Һ����ʱ���ζ�ǰ���ӣ��ζ����ӣ���Һ���ƫС������ҹŨ��ƫ�ͣ�������ȡ����Һ�ĵζ���δ�ô���Һ��ϴ������ҹ��ϡ�ͣ�Ũ��ƫ�ͣ�����ҡ����ƿ�Ĺ����в���������һС������Һ�����ñ�Һ�����ƫС������ҹŨ��ƫ�ͣ��ߵζ����յ�ʱ���ֵζ��ܼ��촦����һ��Һ�Σ���Һ���ƫ����ҹŨ��ƫ�ߣ�ѡC��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| A��NaOH���� | B����NaOH��Һ | C��δ֪Ũ������ | D������ˮ��E��̼������Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ˮú����һ����Դ | B��ˮ���Ƕ�����Դ |
| C����Ȼ���Ƕ�����Դ | D�������Ƕ�����Դ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��

| A����Һ�����Ũ��Ϊ0��1 mol/L |
| B��NaOH��Һ��Ũ��Ϊ0��05 mol/L |
| C���ζ��յ�ʱ�����Ӽ�ʽ�ζ��ܶ���������NaOH��ҺŨ�Ȼ�ƫ�� |
| D��ָʾ����ɫʱ��˵��������NaOHǡ����ȫ��Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| A��c (CH3COO��)��c (Na+)��c (H+)��c (OH��) | B��c (CH3COO��)��c (Na +)��c (OH��)��c (H+) |
| C��c (CH3COO��)��c (H+)��c (Na+)��c (OH��) | D��c (Na+)��c(CH3COO��)��c (OH��)��c (H+) |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����pH=l��CH3COOH��Һ��pH=13��NaOH��Һ�������϶��� |
| B����1mL0��1 mol��L��1CH3COOH��Һ��10 mL 1 mol��L��1��NaOH��Һ��϶��� |
| C����0��1mol��L��1��CH3COOH��Һ��0��1 mol��L��1��NaOH��Һ�������϶��� |
| D����0��1 mol��L��1��CH3COONa��Һ��0��1 mol��L��1��NaOH��Һ�������϶��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������ȼ�����ǽ�ˮ��Ϊ�͵�����ȼ�� |
| B�������Ǿ�����ֵ�ߡ�����Ⱦ���ŵ��ȼ�� |
| C���Ҵ��DZ���������������������ȼ�� |
| D��ʯ�ͺ�ú�ǹ�������ʹ�õĿ������Ļ�ʯȼ��] |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com