


 Сѧ���AB��ϵ�д�
Сѧ���AB��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���϶���SO2��CO2��NO | B���϶�û��O2��NO2 |
| C��������CO��CO2 | D���϶���SO2��NO |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
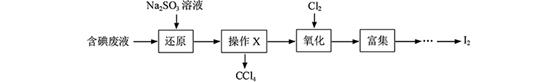
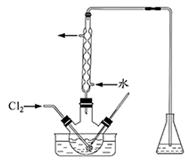
 I2+5SO42��+H2O
I2+5SO42��+H2O�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
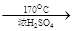 CH2=CH2����H2O��
CH2=CH2����H2O��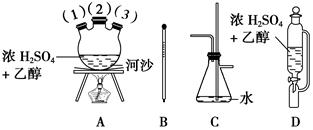
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| ѡ�� | ʵ����� | ʵ������ | ���� |
| A | ij������Һ����AgNO3��Һ | �а�ɫ���� | �����β�һ����NaCl |
| B | Ũ������NaCl�����ϼ��� | ��������� | ��������Ա�HClǿ |
| C | ij��ɫ����ͨ����ˮ�� | ��ˮ��ɫ | ������һ����C2H4 |
| D | ��Ũ�Ⱦ�Ϊ0.1mol��L��1��NaCl��NaI�����Һ�еμ�������Pb(NO3)2��Һ | ���ֻ�ɫ���� ��PbI2�� | KSP��PbI2����KSP��PbCl2�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| ѡ�� | ʵ����� | ʵ������ | ���� |
| A | ȡ�����ʵ��������ֽ�������X��Y���ֱ������������ᷴӦ | X���������������Y�� | �����ԣ�X>Y |
| B | �����pH��3��HA��HB������ֱ���������п��Ӧ����ˮ���ռ����� | HA�ų����������ҷ�Ӧ���ʿ� | HA���Ա�HBǿ |
| C | ��CuSO4��Һ�м���KI��Һ���ټ��뱽���� | �а�ɫ�������ɣ��������ɫ | ��ɫ��������ΪCuI |
| D | ȡ���õ�Na2O2��ĩ�������еμӹ��������� | ������ɫ���� | Na2O2û�б��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
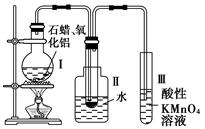
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��������Ʒ�����ȡ���ȴ������CuO |
| B��������Ʒ�����ȡ���ȴ������Cu(NO3)2 |
| C��������Ʒ�����ȡ�����֪��������ˮ�Ȼ�������ˮ���������� |
| D��������Ʒ����NaOH�����ˡ����ȡ���ȴ������CuO |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com