
 ҪтЗЕҪМУэјЖЛгРЎЧҙФӘПөБРҙр°ё
ҪтЗЕҪМУэјЖЛгРЎЧҙФӘПөБРҙр°ё
| Дкј¶ | ёЯЦРҝОіМ | Дкј¶ | іхЦРҝОіМ |
| ёЯТ» | ёЯТ»Гв·СҝОіМНЖјцЈЎ | іхТ» | іхТ»Гв·СҝОіМНЖјцЈЎ |
| ёЯ¶ю | ёЯ¶юГв·СҝОіМНЖјцЈЎ | іх¶ю | іх¶юГв·СҝОіМНЖјцЈЎ |
| ёЯИэ | ёЯИэГв·СҝОіМНЖјцЈЎ | іхИэ | іхИэГв·СҝОіМНЖјцЈЎ |
ҝЖДҝЈәёЯЦР»ҜС§ АҙФҙЈә МвРНЈә
ЎпЎпЎп
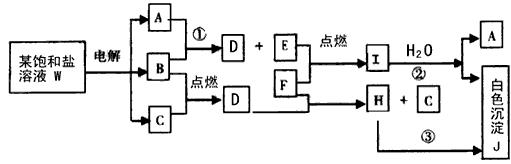
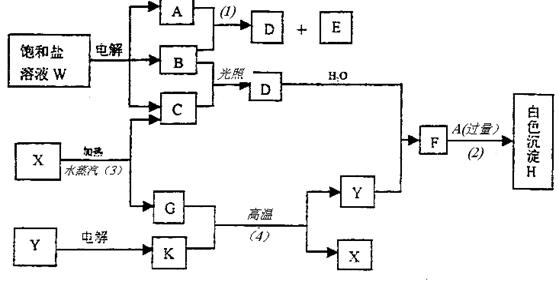
ұҘәНСОИЬТәWөДөзҪвІъОп·ўЙъПВБРПөБР·ҙУҰЎЈНјЦРөДГҝТ»·ҪёсұнКҫУР№ШөДТ»ЦЦЦчТӘ·ҙУҰОп»тЙъіЙОпЈЁ·ҙУҰЦРјУИл»тЙъіЙөДЛ®ТФј°ЙъіЙөДЖдЛьІъОпТСВФИҘЈ©Ј¬ЖдЦРAЎўBЎўCЎўDЎўEФЪіЈОВПВҫщОӘёЯЦР»ҜС§ЦРіЈјыЖшМ¬ОпЦКЈ¬XәНKКЗЦРС§іЈјыөДҪрКфөҘЦКЈ¬Сх»ҜОпYКЗТ»ЦЦұИҪПәГөДДН»рІДБПЎЈHҝЙИЬУЪЗҝЛбЗҝјоЎЈ
»ШҙрПВБРОКМвЈә
ўЩWөДГыіЖКЗ Ј¬GөДЛЧГыКЗ ЎЈ
ўЪA·ЦЧУөДҝХјд№№РНКЗ ЎЈ
ўЫ·ҙУҰЈЁ3Ј©өД»ҜС§·ҪіМКҪКЗ ЎЈ
·ҙУҰЈЁ4Ј©өД»ҜС§·ҪіМКҪКЗ ЎЈ
ўЬұҘәНСОИЬТәWөзҪвөДАлЧУ·ҪіМКҪКЗ ЎЈ
ўЭТСЦӘCөДИјЙХИИОӘ285.8kJ/molЎЈКФРҙіцCНкИ«ИјЙХЙъіЙТәМ¬ОпЦКөДИИ»ҜС§·ҪіМКҪ ЎЈ
Ійҝҙҙр°ёәНҪвОц>>
№ъјКѧУУЕСЎ - Б·П°ІбБРұн - КФМвБРұн
әюұұКЎ»ҘБӘНшОҘ·ЁәНІ»БјРЕПўҫЩұЁЖҪМЁ | НшЙПУРәҰРЕПўҫЩұЁЧЁЗш | өзРЕХ©ЖӯҫЩұЁЧЁЗш | ЙжАъК·РйОЮЦчТеУРәҰРЕПўҫЩұЁЧЁЗш | ЙжЖуЗЦИЁҫЩұЁЧЁЗш
ОҘ·ЁәНІ»БјРЕПўҫЩұЁөз»°Јә027-86699610 ҫЩұЁУКПдЈә58377363@163.com