��2013?�ߺ�ģ�⣩A��B��C��D��E��F�����ֶ���������Ԫ�أ����ǵ�ԭ������������������A��D��C��F�ֱ���ͬһ����Ԫ�أ�A��F��Ԫ�ص�ԭ�Ӻ���������֮�ͱ�C��D��Ԫ��ԭ�Ӻ���������֮����2��FԪ�ص������������Ǵ�����������0.75������֪BԪ�ص��������������ڲ��������2����EԪ�ص�������������������Ӳ�������ش�
��1��1mol��E��F����Ԫ����ɵĻ��������A��C��D����Ԫ����ɵĻ��������Ӧ����ȫ��Ӧ�����ĺ��ߵ����ʵ���Ϊ
8mol
8mol
��
��2��A��C��F����γɼס��������������Ǿ�Ϊ��һ��˫ԭ�������ӣ�����18�����ӣ�����10�����ӣ�������ҷ�Ӧ�����ӷ���ʽΪ
HS-+OH-=S2-+H2O
HS-+OH-=S2-+H2O
��
��3������B��ȼ����akJ/mol����B��C����Ԫ����ɵĻ�����BC14g��ȫȼ�շų�bkJ������д������B�͵���C��Ӧ����BC���Ȼ�ѧ����ʽ��
C��s��+
O
2��g��=CO��g������H=-��a-2b��kJ/mol
C��s��+
O
2��g��=CO��g������H=-��a-2b��kJ/mol
��
��4����ҵ���ڸ��µ������£�������A
2C��BC��Ӧ��ȡ����A
2���ڵ�����Ģ������ܱ������зֱ����1molA
2C��1molBC��2molA
2C��2molBC��һ�������£���ַ�Ӧ��ֱ�ﵽƽ�⣨�������¶���ȣ�������˵����ȷ����
AB
AB
A���ﵽƽ������Ҫ��ʱ�䣺��
B���ﵽƽ���A
2C��ת���ʣ���=��
C���ﵽƽ���BC�����ʵ�������
D���ﵽƽ���A
2�������������
E���ﵽƽ������ջ�ų�����������=��
F���ﵽƽ�����ϵ��ƽ����Է�����������
��5����BԪ�صĵ�����EԪ�صĵ��ʿ����Ƴɵ缫������A��C��D����Ԫ����ɵĻ��������Һ�й��ɵ�أ����ظ����ĵ缫��Ӧʽ��
Al-3e-+4OH-=AlO2-+2H2O
Al-3e-+4OH-=AlO2-+2H2O
��




 ���ɶ���ܲ��¿�ֱͨ�п�ϵ�д�
���ɶ���ܲ��¿�ֱͨ�п�ϵ�д�






 ��
�� ��
�� ��
��
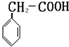 ��
�� ��
�� ��
��