
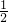 ��0.1mol/L=0.05mol/L��
��0.1mol/L=0.05mol/L��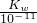 mol/L������KW=1.0��10-13��
mol/L������KW=1.0��10-13��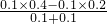 =0.1mol/L����C��H+��=
=0.1mol/L����C��H+��=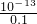 mol/L=10-12 mol/L���ʷ�Ӧ����Һ��pH=-lg10-12=12��
mol/L=10-12 mol/L���ʷ�Ӧ����Һ��pH=-lg10-12=12��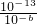 ����a+b=12����pHa+pHb=12��
����a+b=12����pHa+pHb=12��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013���㽭ʡ��Ҧ���и����ڶ����¿���ѧ�Ծ����������� ���ͣ������
��12�֣���1��ij�о���ѧϰС����ʵ����������1 mol/L��ϡ�������Һ��Ȼ������ζ�ijδ֪Ũ�ȵ�NaOH��Һ�������й�˵������ȷ����______________��
A��ʵ�������õ��ĵζ��ܡ�����ƿ����ʹ��ǰ����Ҫ��©��
B�����ʵ��������60 mL ��ϡ�������Һ������ʱӦѡ��100 mL����ƿ��
C������ƿ�к�����������ˮ���ᵼ���������Һ��Ũ��ƫС��
D����ʽ�ζ���������ˮϴ�Ӻ�װ���Ũ�ȵ�ϡ���ᣬ���õ�NaOH��Һ��Ũ�Ƚ�ƫ��
E��������Һʱ���������һ�ζ���ʱ���Ӷ������������ʵ����ƫ��
F���к͵ζ�ʱ���������һ�ζ���ʱ���Ӷ������������ʵ����ƫ��
��2�������£���֪0.1 mol��L��1һԪ��HA��Һ��c(OH��) / c(H+)��1��10��8��
�ٳ����£�0.1 mol��L��1 HA��Һ��pH= ��д�����ᣨHA����NaOH��Һ��Ӧ�����ӷ���ʽ�� ��
��pH��3��HA��pH��11��NaOH��Һ�������Ϻ���Һ��4���������ʵ���Ũ�ȴ�С��ϵ�ǣ� ��
��0.2 mol��L��1HA��Һ��0.1mol��L��1NaOH��Һ�������Ϻ�������Һ�У�
c(H+)��c(HA)��c(OH��)�� mol��L��1������Һ����仯���Բ��ƣ�
��3��t��ʱ����pH=2��ϡ�����pH=11��NaOH��Һ�������Ϻ���Һ�����ԣ�����¶���ˮ�����ӻ�����Kw= ��
�ٸ��¶��£�t�棩����100 mL 0.1 mol��L-1��ϡH2SO4��Һ��100 mL 0.4 mol��L-1��NaOH��Һ��Ϻ���Һ����仯���Բ��ƣ�����Һ��pH= ��
�ڸ��¶��£�t�棩��1�����ϡ�����10�����NaOH��Һ��Ϻ���Һ�����ԣ���ϡ�����pH��pHa����NaOH��Һ��pH��pHb���Ĺ�ϵ�ǣ� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ���㽭ʡ�����ڶ����¿���ѧ�Ծ��������棩 ���ͣ������
��12�֣���1��ij�о���ѧϰС����ʵ����������1 mol/L��ϡ�������Һ��Ȼ������ζ�ijδ֪Ũ�ȵ�NaOH��Һ�������й�˵������ȷ����______________��
A��ʵ�������õ��ĵζ��ܡ�����ƿ����ʹ��ǰ����Ҫ��©��
B�����ʵ��������60 mL ��ϡ�������Һ������ʱӦѡ��100 mL����ƿ��
C������ƿ�к�����������ˮ���ᵼ���������Һ��Ũ��ƫС��
D����ʽ�ζ���������ˮϴ�Ӻ�װ���Ũ�ȵ�ϡ���ᣬ���õ�NaOH��Һ��Ũ�Ƚ�ƫ��
E��������Һʱ���������һ�ζ���ʱ���Ӷ������������ʵ����ƫ��
F���к͵ζ�ʱ���������һ�ζ���ʱ���Ӷ������������ʵ����ƫ��
��2�������£���֪0.1 mol��L��1һԪ��HA��Һ��c(OH��) / c(H+)��1��10��8��
�������£�0.1 mol��L��1 HA��Һ��pH= ��д�����ᣨHA����NaOH��Һ��Ӧ�����ӷ���ʽ�� ��
��pH��3��HA��pH��11��NaOH��Һ�������Ϻ���Һ��4���������ʵ���Ũ�ȴ�С��ϵ�ǣ� ��
��0.2 mol��L��1HA��Һ��0.1mol��L��1NaOH��Һ�������Ϻ�������Һ�У�
c(H+)��c(HA)��c(OH��)�� mol��L��1������Һ����仯���Բ��ƣ�
��3��t��ʱ����pH=2��ϡ�����pH=11��NaOH��Һ�������Ϻ���Һ�����ԣ�����¶���ˮ�����ӻ�����K w= ��
�ٸ��¶��£�t�棩����100 mL 0.1 mol��L-1��ϡH2SO4��Һ��100 mL 0.4 mol��L-1��NaOH��Һ��Ϻ���Һ����仯���Բ��ƣ�����Һ��pH= ��
�ڸ��¶��£�t�棩��1�����ϡ�����10�����NaOH��Һ��Ϻ���Һ�����ԣ���ϡ�����pH��pHa����NaOH��Һ��pH��pHb���Ĺ�ϵ�ǣ� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ���㽭ʡ�����а�У�����������ϣ���ĩ��ѧ�Ծ��������棩 ���ͣ������
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com