
��֪ijһ��ȼ�Ϻ�̼�����������Ԫ�أ�Ϊ�ⶨ��ȼ�ϵ���ɣ�����ȼ�Ϸ��뵽����������ȼ�գ���ʹ������CO2��H2O�����Լ�ʣ���O2ȫ��ͨ������ͼ��ʾ��װ�ã��õ����±����г���ʵ�����ݣ��������ɵ�������ȫ�������գ���
| | ʵ��ǰ | ʵ��� |
| ������ / g | 101��1 | 103��8 |
| �ҵ����� / g | 82��0 | 86��4 |
1:3��C2H6O
���������������1���������װ��֪��Ũ������������ˮ��������װ�����ӵ�����ΪH2O��������m(H2O)=103��8g-101��1g=2��7g��n(H2O)=0.15mol��n(H)=0.3mol����ʯ����������CO2����װ�����ӵ�����ΪCO2��������m(CO2)=86��4g-82��0g=4��4g��n(C)=n(CO2)=0.1mol��̼����ԭ�ӵ���Ŀ��Ϊ1:3����2����÷��Ӻ���n��̼ԭ�ӣ�����ԭ��Ϊ3n������Է�������Ϊ12n+3n+16=46����n=2���÷�����2��̼ԭ�ӣ�6����ԭ�Ӻ�һ����ԭ�ӣ��ʸ����ʵķ���ʽΪC2H6O��
���㣺�����л������ʽ��ȷ����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
���ȩ��һ��ʳƷ���Ӽ�����������ľ����ԭ�Ϻϳɣ��ϳ�·������ͼ��ʾ������˵����ȷ���ǣ� �� 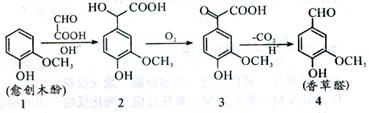
| A�������Ϸ�Ӧ1��2��ԭ��������Ϊ100% |
| B��������2���ܷ������۷�Ӧ |
| C�������Ƶõ����ȩ���Ƿ���л�����3�������Ȼ�����Һ |
| D�������ʵ��������ֻ�����ֱ�������NaOH��Ӧ������NaOH���ʵ���֮��Ϊ1��3��2��4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��8�֣����������dz����ڵ��ƾ��в�ݮ�����ѡ�ӣ�ҡ�����������ζ��ʳ���㾫�����ķ���ʽΪC10H10O2���ҷ�����ֻ����1��������������ֻ��һ��ȡ��������֪ijһ�����������ķ��ӽṹģ������ͼ��ʾ��ͼ��������֮�����߱�ʾ������˫������
�Իش��������⡣
���������Ľṹ��ʽΪ ����2�������еĹ����ŵ�����Ϊ ��
��3���ڱ����ϵ�һ�ȴ����� �֡�
��4��д������������������������Һ��ˮ��Ļ�ѧ����ʽ��_________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��10�֣����ȡ���� ���Ա�KMnO4������Һ������
���Ա�KMnO4������Һ������ ���������R��ֱ���뱽��������̼ԭ����û��C��H�������ױ������õ�
���������R��ֱ���뱽��������̼ԭ����û��C��H�������ױ������õ� �����з���ʽ��C11H16��һ���ȡ��������֪�����Ա�������
�����з���ʽ��C11H16��һ���ȡ��������֪�����Ա������� ���칹�干��7�֣�����������
���칹�干��7�֣����������� ��
��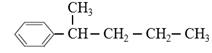 ��
��
��д������5�ֵĽṹ��ʽ��_____________��______________��__________________��_____________��____________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��16�֣�
��I��������Ӧ�У�����������Ӧ����_______����ȡ����Ӧ����_______���ڼӳɷ�Ӧ����_______������ţ�
���Ҵ��������ظ������Һ�ķ�Ӧ
�������ڹ�����������������Ӧ
����ϩʹ���Ը��������Һ��ɫ
����ϩʹ��ˮ��ɫ
�ݱ���Ũ����Ũ�����Ϲ�����ȡ������
����֬������������µ�ˮ�ⷴӦ
�߱���һ���������������ķ�Ӧ
��II��ƻ���� 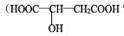 ��������ˮ���ǹ������Ӽ�
��������ˮ���ǹ������Ӽ�
��1��д��ƻ�����й����ŵ�����_____________________
��2��д��ƻ����ֱ����������ʷ�Ӧ�Ļ�ѧ����ʽ���л����ýṹ��ʽ��ʾ��
����Na�ķ�Ӧ______________________________________________
����Na2CO3�ķ�Ӧ__________________________________________
��III��д��ʵ������ת���Ļ�ѧ����ʽ���л����ýṹ��ʽ��ʾ��
�Ҵ��Ĵ�����_______________________________________________
����������___________________________________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��Ȼά����P���ṹ����ͼ�����ӽṹ��RΪ���������������ڻ��������У�����һ��Ӫ��������������ά����P��������ȷ���� 
| A��������ˮ��Ӧ����1 mol��������������ˮ��Ӧ����6 mol Br2 |
| B������NaOH��Һ��Ӧ��1 mol�����ʿ���5 mol NaOH��Ӧ |
| C��һ��������1 mol�����ʿ���H2�ӳɣ���H2�����Ϊ6 mol |
| D��ά����P�ܷ���ˮ�ⷴӦ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪����A����ʽΪC3H4O2�������ԡ�FΪ���߸�ԭ����ɵĻ�״�ṹ������ʽΪC6H8O4����������¿�ͼ�ش����⣺
��1��A�Ľṹ��ʽΪ________________________________��
��2����Ӧ�ٵķ�Ӧ����Ϊ__________________________��
��3��������B�к��������ŵ�������______________________________��
��4��D��E����F�Ļ�ѧ����ʽ__________________________________��
D��E����1:1��ӦҲ�����ɸ߾����д�����ɸø߾���Ļ�ѧ��Ӧ����ʽ=______
________________________________________________________________________��
��5��G����H�Ļ�ѧ����ʽ________________________________________________��
��6��д��C��ͬ���칹���������������ʵĽṹ��ʽ_______________________��
________________________��____________________________________������д3����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(14��)��ij�������Ļ�����A������Է�������Ϊ104��̼����������Ϊ92��3%��
(1) A��������Ȼ�̼��Һ��Ӧ�Ļ�ѧ����ʽΪ ;
(2)��֪


��д��A��ϡ�����KMnO4��Һ�ڼ��������·�Ӧ���ò������������ᷴӦ�Ļ�ѧ����ʽ�� ;
(3)��һ�������£���A�ۺϵõ��ĸ߷��ӻ�����Ľṹ��ʽΪ������ ����
(4) A��������ȫ�ӳɺ��һ�ȴ��ﹲ���������֡�
������ͼ��ʾװ����ȡ������������ش��������⡣ 
(5)���Թ�������һ���������Ҵ��������ŨH2SO4�Ļ��Һ�ķ�����_______
(6)ĩ�˸���ܵ�������_______________��
(7)ʵ�������ȡ��ʢ�б���̼������Һ���Թܣ����ظ��Թ��ڱڻ���������ɫʯ����Һ1����������ʯ���Ϊ���㻷�����϶�������ɫΪ_________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ѧ����ѡ��5���л���ѧ������ ��15�֣�
��ͼ�� A��B��C��D��E��Ϊ�л��������֪��C�ܸ�NaHCO3������Ӧ���ú�D����Է���������ȣ���EΪ��֧���Ļ����
������ͼ�ش����⣺
��1����֪E����Է�������Ϊ102������̼��������Ԫ�ص����������ֱ�Ϊ58.8%��9.8%������Ϊ����C�����еĹ����������� ______________��������B���ܷ����ķ�Ӧ�ǣ�����ĸ��ţ���______________
a���ӳɷ�Ӧ b��ȡ����Ӧ c����ȥ��Ӧ
d��������Ӧ e��ˮ�ⷴӦ f���û���Ӧ
��2����Ӧ�ڵĻ�ѧ����ʽ��__________________��
��3����Ӧ��ʵ���м��ȵ�Ŀ���ǣ� .��
��4��A�Ľṹ��ʽ�� __________________��
��5��д��ͬʱ������������������B��ͬ���칹������ͬ���칹��Ľṹ��ʽ��
���м��ȡ�������ṹ
�����ڷ������γɵ���
���� FeCl3��Һ������ɫ��Ӧ
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com