
����Ĺ�ҵ�Ʊ���һ����Ҫ�Ļ����������̣���ͬʱ�����������л��������SO2����Ⱦ�
��1����SO2ͨ��Fe(NO3)3��Һ�У���Һ���ػ�ɫ��Ϊdz��ɫ���������ֱ�Ϊ�ػ�ɫ����ʱ������BaCl2��Һ����������ɫ��������Һ���ػ�ɫ��Ϊdz��ɫ�����ӷ���ʽ��ʾΪ_____��������dz��ɫ��Ϊ�ػ�ɫ�����ӷ���ʽΪ_____��
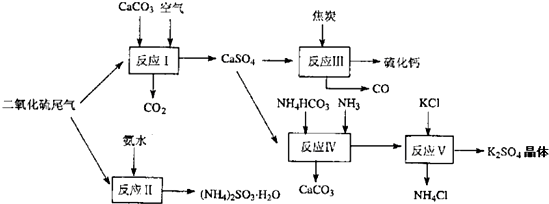
��2�������Ṥҵ��β������ˮ��ʯ��ʯ����̿��̼����狀�KCIΪԭ�Ͽ��Ժϳ�����Ҫ��;���ơ�����ء���������淋����ʡ��ϳ�·�����£�
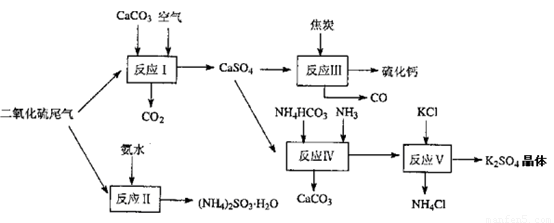
д����Ӧ��Ӧ���Ļ�ѧ����ʽ ��
��ӦIII���������뻹ԭ�������ʵ���֮��Ϊ__ __��
��ӦV��25'C��40%�Ҷ�����Һ�н��У��ø��ֽⷴӦ��˳�����е�ԭ���� ��
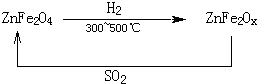
��3���������ײ�����ȱλ����п��ZnFe2Ox������������ʹSO2�ֽ⣬��С��ҵ�����Ի�����Ӱ�죬��������п��ZnFe2O4�������»�ԭ�Ƶã�ת����������ͼ��ʾ��
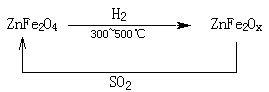
��2molZnFe2Ox��SO2��Ӧ������ 0.75molS��x�� ,д������п�����±���ԭ������ȱλ����п�Ļ�ѧ����ʽ
��4��ʯ��ʯ-ʯ��ʪ�����������ռ�����ԭ���������еĶ��������뽬Һ�е�̼����Լ�������Ӧ����ʯ�ࣨCaSO4.2H2O����д���÷�Ӧ�Ļ�ѧ����ʽ ��ij�糧��ú300t��ú�к������������Ϊ2.5%������ȼ��ʱú�е���ȫ��ת��Ϊ���������ø÷�������ʱ��96%����ת��Ϊʯ�࣬�������ʯ�� t��
����16�֣���1��SO2 + 2Fe3+ + 2H2O��SO42- + 2Fe2+ + 4H+��2�֣�
3Fe2+ + NO3- + 4H+��3Fe3+ + NO��+ 2H2O��2�֣�
��2��NH4HCO3 + CaSO4 + NH3��CaCO3��+ (NH4)2SO4 ��2�֣� 1:4��1�֣�
K2SO4���Ҷ�����Һ�е��ܽ��С��1�֣�
��3��3.25��2�֣� 4ZnFe2O4
+ 3H2 4ZnFe2O3.25
+ 3H2O��2�֣�
4ZnFe2O3.25
+ 3H2O��2�֣�
��4��2CaSO4 + 2SO2 + O2 + 4H2O��2��CaSO4.2H2O��+2CO2 ��2�֣� 38.7��2�֣�
��������
�����������1��SO2���л�ԭ�ԣ������Ӿ��������ԣ��������ܰ�SO2�����������ᣬ�������ӱ���ԭΪ�������ӣ�������Һ���ػ�ɫ��Ϊdz��ɫ����Ӧ�����ӷ���ʽ��SO2 + 2Fe3+ + 2H2O��SO42- + 2Fe2+ + 4H+�����������������£�NO3������ǿ�����ԣ��ܰ����������������������ӣ��������dz��ɫ��Ϊ�ػ�ɫ����Ӧ�����ӷ���ʽΪ3Fe2+ + NO3- + 4H+��3Fe3+ + NO��+ 2H2O��
��2����Ӧ���ķ�Ӧ��������ơ�̼����狀Ͱ�����������֮һ��̼��ƣ������ԭ���غ��֪������һ��������������泥���˷�Ӧ�Ļ�ѧ����ʽ��NH4HCO3 + CaSO4 + NH3��CaCO3��+ (NH4)2SO4����Ӧ����̼�Ļ��ϼ۴�0�����ߵ���2�ۣ�ʧȥ2�����ӣ�����������Ӧ��̼�ǻ�ԭ����SԪ�صĻ��ϼ۴ӣ�6�۽��͵���2�ۣ��õ�8�����ӣ�������ԭ��Ӧ���������������������Ӧ���ݵ��ӵĵ�ʧ�غ��֪���ڷ�Ӧ���������뻹ԭ�������ʵ���֮��Ϊ1:4������K2SO4���Ҷ�����Һ�е��ܽ��С���ܹ��γɾ������������˷��ϸ��ֽⷴӦ������������
��3��ZnFe2OX��Fe��ƽ����̬Ϊ����x��1�������������������Ļ��ϼ��ǣ�3�ۣ������ڷ�Ӧ��ʧȥ��3��x��1�������ӡ���SO2��S ��S�Ļ��ϼ۴ӣ�4�۽��͵�0�ۣ��õ�4�����ӣ����Ը��ݵ��ӵĵ�ʧ�غ��֪��2mol��2����3��x��1����0.75��4�����x��3.25����������п�����±���ԭ������ȱλ����п�Ļ�ѧ����ʽΪ4ZnFe2O4
+ 3H2 4ZnFe2O3.25
+ 3H2O��
4ZnFe2O3.25
+ 3H2O��
��4������ԭ���غ��֪����Ӧ�л�Ӧ����CO2���ɣ���˷�Ӧ�Ļ�ѧ����ʽ��2CaSO4 + 2SO2 + O2 + 4H2O��2��CaSO4.2H2O��+2CO2 ������ԭ���غ��֪��
S������������CaSO4��2H2O
32t 172t
300t��2.5%��96% m
���m��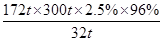 ��38.7t
��38.7t
���㣺����SO2�������Լ������Ӻ��������ӵ����ʣ�������ԭ��Ӧ����ʽ����д�Լ��йؼ��㣻��Ӧ�����Ŀ��ƣ����ݷ���ʽ���е��йؼ����


 53���ò�ϵ�д�
53���ò�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��2012?��ͷ��ģ���״���Դ�ḻ���۸�������������淽�㣬��һ����Ҫ�Ļ���ԭ�ϣ�������Ҫ����;��Ӧ��ǰ����
��2012?��ͷ��ģ���״���Դ�ḻ���۸�������������淽�㣬��һ����Ҫ�Ļ���ԭ�ϣ�������Ҫ����;��Ӧ��ǰ����| 1 |
| 2 |
| ���� |
| ���� |
| ���� |
| ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ����ѧ��ѧʵ���У�ͨ������ˮ����ͭ��������ˮ�Ĵ��ڣ�������ˮ����ͭ��ʪ�Ժ�ǿ����Ҫ�������ã�
����ѧ��ѧʵ���У�ͨ������ˮ����ͭ��������ˮ�Ĵ��ڣ�������ˮ����ͭ��ʪ�Ժ�ǿ����Ҫ�������ã�
| ||
| ||
| ||
| ||


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��ӱ�ʡʯ��ׯ�б�ҵ���ѧ������⣨�������ۻ�ѧ�Ծ��������棩 ���ͣ������
����Ĺ�ҵ�Ʊ���һ����Ҫ�Ļ����������̣��������������л����������Ⱦ����Ҫ�����������п��ǵ���ɫ���ա�
Iβ�������պ��ۺ����á�
�Թ�ҵ�������β������ˮ��ʯ��ʯ����̿��̼����狀�KCIΪԭ�Ͽ��Ժϳ��ơ�����ء�������淋����ʡ��ϳ�·�����£�
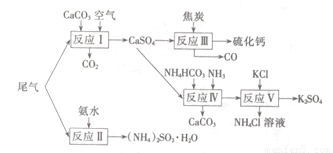
��1����ӦIII���������뻹ԭ�������ʵ���֮��Ϊ???????????? ��
��2����Ӧ���Ļ�ѧ����ʽΪ?????????????????????? ��
��3����ӦV��25�桢40%���Ҷ�����Һ�н��У��÷�Ӧ��˳�����е�ԭ��Ϊ??????????? ��
������Ļ������á�
SO2�Ĵ�������ʹ�õĴ���ΪV2O5��ʵ�������У�������ʹ��һ��ʱ��Ậ��V2O5��VOSO4��SiO2�ȣ�����VOSO4��������ˮ������V2O5������Ҫ�������£�
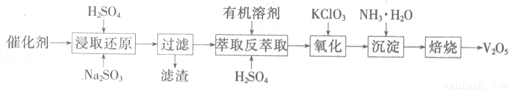
��4��������ȡʹ�õ�������������һ������ʱ������____??????? ��������
��5����ȡ��ԭ���̵IJ���֮һ��VOSO4����Ӧ�Ļ�ѧ����ʽΪ?????????????????? ��
�������̵Ļ�ѧ����ʽΪKClO3+6VOSO4+3H2SO4= 2(VO)2(SO4)3+KCl+3H2O�������������Լ�Na2SO3��KC1O3�����ʵ���֮��Ϊ12��7����ô�����V2O5��VOSO4�����ʵ���֮��Ϊ??????????????? ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com