
 l3��Һ�ܽ�Ag�ķ�Ӧ�������ȫ���������ԭ����������������������������
l3��Һ�ܽ�Ag�ķ�Ӧ�������ȫ���������ԭ����������������������������| ʵ�鲽�裨��Ҫ��д����������̣� | Ԥ������ͽ��� |
| �� �� ���� | ��������ʧ������2������ ����������ʧ������2�������� |
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| ʵ�� ��� | ���/mL | ||||
| K2S2O8��Һ | ˮ | KI��Һ | Na2S2O3��Һ | ������Һ | |
| �� | 10.0 | 0.0 | 4.0 | 4.0 | 2.0 |
| �� | 9.0 | 1.0 | 4.0 | 4.0 | 2.0 |
| �� | 8.0 | VX | 4.0 | 4.0 | 2.0 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| ���� | ��ʼ����ʱ��pH | ��ȫ����ʱ��pH |
| Fe2�� | 6��34 | 9��7 |
| Fe3�� | 1��48 | 3��2 |
| Zn2�� | 6��2 | 8��0 |
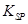 = 2��70��10-39]
= 2��70��10-39]�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
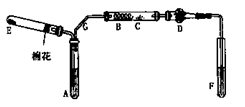
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 l2����Ư���ԡ�
l2����Ư���ԡ�
 ��
�� ��ɫ����������ɵó��Ľ�����
��ɫ����������ɵó��Ľ����� �鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
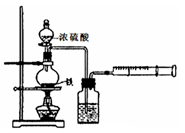
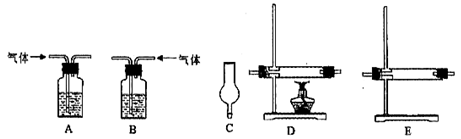
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
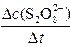 ��
��| ʵ �� �� �� | �� | �� | �� | �� | �� | |
| �Լ� ���� (mL) | 0.20 mol��L-1(NH4)2S2O8��Һ | 20.0 | 10.0 | b | 20.0 | 20.0 |
| 0.20 mol��L-1 KI��Һ | 20.0 | 20.0 | 20.0 | 10.0 | 5.0 | |
| 0.010 mol��L-1 Na2S2O3��Һ | a | 8.0 | 8.0 | 8.0 | 8.0 | |
| 0.2% ������Һ | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 | |
| 0.20 mol��L-1KNO3��Һ | 0 | 0 | 0 | 10.0 | c | |
| 0.20 mol��L-1(NH4)2SO4��Һ | 0 | 10.0 | 15.0 | 0 | 0 | |
| 20 ��ʱ����Ӧʱ�䦤t��s�� | 32 | 67 | 130 | 66 | 135 | |
| Ϊ��ʹ��Һ������ǿ�Ⱥ���������ֲ��䣬���ٵ�(NH4)2S2O8��Һ��KI��Һ���������ֱ���(NH4)2SO4��Һ��KNO3��Һ���㣻��Һ��Ϻ�������� | ||||||
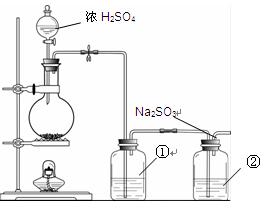
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| ʵ�鷽�� ��ֻ��дѡ���Լ���ţ� | ʵ������ | ���� | |
| �� | | һ֧�Թ����а�ɫ�������ɣ���һ֧�Թ��������ɰ�ɫ���������ɫ������ʧ | �����ԣ� |
| �� | |  | �ǽ����ԣ�Cl��Br |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com