

 Na2SiO3ЃЋCO2ЁќЃЛ
Na2SiO3ЃЋCO2ЁќЃЛ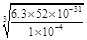 ЃЈЛђ
ЃЈЛђ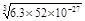 Лђ
Лђ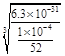 ЃЛЃЉ
ЃЛЃЉ Na2SiO3ЃЋCO2ЁќЁЃ(4)CH3OHЁЂH2ЕФШМЩеЕФШШЛЏбЇЗНГЬЪНЮЊЃКЂй CH3OH(l)+3/2O2(g)=CO2(g)+2H2O(l) ІЄH=Ѓ725.5 kJ/mol ; ЂкH2(g)+ 1/2O2(g)=H2O(l) ІЄH=Ѓ285.8 kJ/mol. ЂкЁС3ЃЂйЁЃећРэПЩЕУCO2(g)ЃЋ3H2(g) = CH3OH(l)ЃЋH2O(l) ІЄH=Ѓ131.9 kJ/molЃЛЃЈ5ЃЉЂйИљОнЕчзгЪиКуЁЂЕчКЩЪиКуМАжЪСПЪиКуЖЈТЩПЩЕУЕчНтКЌCr2O72-ЕФЫсадЗЯЫЎЕФзмЗДгІЕФРызгЗНГЬЪНЮЊЃК6FeЃЋCr2O72-ЃЋ2H+ЃЋ17H2O = 6Fe(OH)3Ё§ЃЋ2Cr(OH)3Ё§ЃЋ6H2ЁќЃЛЂкШєЕиБэЫЎИѕКЌСПзюИпЯожЕЪЧ0.1 mg/L ЃЌc(Cr3+) =1ЁС10-4gЁТ52g/mol/L=
Na2SiO3ЃЋCO2ЁќЁЃ(4)CH3OHЁЂH2ЕФШМЩеЕФШШЛЏбЇЗНГЬЪНЮЊЃКЂй CH3OH(l)+3/2O2(g)=CO2(g)+2H2O(l) ІЄH=Ѓ725.5 kJ/mol ; ЂкH2(g)+ 1/2O2(g)=H2O(l) ІЄH=Ѓ285.8 kJ/mol. ЂкЁС3ЃЂйЁЃећРэПЩЕУCO2(g)ЃЋ3H2(g) = CH3OH(l)ЃЋH2O(l) ІЄH=Ѓ131.9 kJ/molЃЛЃЈ5ЃЉЂйИљОнЕчзгЪиКуЁЂЕчКЩЪиКуМАжЪСПЪиКуЖЈТЩПЩЕУЕчНтКЌCr2O72-ЕФЫсадЗЯЫЎЕФзмЗДгІЕФРызгЗНГЬЪНЮЊЃК6FeЃЋCr2O72-ЃЋ2H+ЃЋ17H2O = 6Fe(OH)3Ё§ЃЋ2Cr(OH)3Ё§ЃЋ6H2ЁќЃЛЂкШєЕиБэЫЎИѕКЌСПзюИпЯожЕЪЧ0.1 mg/L ЃЌc(Cr3+) =1ЁС10-4gЁТ52g/mol/L=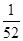 ЁС10-4 mol/L .Cr(OH)3ЕФKsp=6.3ЁС10ЈC31ЃЌМДc(Cr3+)ЁЄc3(OH-)Ён6.3ЁС10ЈC31;
ЁС10-4 mol/L .Cr(OH)3ЕФKsp=6.3ЁС10ЈC31ЃЌМДc(Cr3+)ЁЄc3(OH-)Ён6.3ЁС10ЈC31;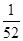 ЁС10-4=6.3ЁС52ЁС10-27.Ыљвдc(OH-)=
ЁС10-4=6.3ЁС52ЁС10-27.Ыљвдc(OH-)=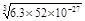 .
.

 дФЖСПьГЕЯЕСаД№АИ
дФЖСПьГЕЯЕСаД№АИ
| ФъМЖ | ИпжаПЮГЬ | ФъМЖ | ГѕжаПЮГЬ |
| ИпвЛ | ИпвЛУтЗбПЮГЬЭЦМіЃЁ | ГѕвЛ | ГѕвЛУтЗбПЮГЬЭЦМіЃЁ |
| ИпЖў | ИпЖўУтЗбПЮГЬЭЦМіЃЁ | ГѕЖў | ГѕЖўУтЗбПЮГЬЭЦМіЃЁ |
| ИпШ§ | ИпШ§УтЗбПЮГЬЭЦМіЃЁ | ГѕШ§ | ГѕШ§УтЗбПЮГЬЭЦМіЃЁ |
ПЦФПЃКИпжаЛЏбЇ РДдДЃКВЛЯъ ЬтаЭЃКЬюПеЬт


ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃКВЛЯъ ЬтаЭЃКЕЅбЁЬт
| | ЪЕбщФПЕФ | ЪЕбщВйзї |
| A | жЄУїввЯЉФмЗЂЩњМгГЩЗДгІ | НЋввЯЉЦјЬхЭЈШыфхЕФЫФТШЛЏЬМШмвКжа |
| B | МјБ№Fe(OH)3НКЬхКЭFeCl3ШмвК | гУМЄЙтБЪЗжБ№ееЩфСНжжЮяжЪЃЌДгВрУцЙлВьЪЧЗёГіЯжЙтССЕФЭЈТЗЁЃ |
| C | МьбщОЦОЋжаЪЧЗёКЌгаЫЎ | ШЁЩйСПОЦОЋМгШыЮоЫЎСђЫсЭЗлФЉ |
| D | МјБ№БНЁЂЛЗвбЯЉКЭвбЭщ | МгШыЫсадИпУЬЫсМиШмвК |
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃКВЛЯъ ЬтаЭЃКЕЅбЁЬт
| бЁЯю | ЮяжЪ(РЈКХжаЮЊдгжЪ) | Г§дгЪдМС | Вйзї |
| A | ЕэЗл(NaCl) | ЫЎ | Й§ТЫ |
| B | BaCO3(BaSO4) | БЅКЭNa2CO3ШмвК | НСАшЁЂЙ§ТЫ |
| C | ввЫсввѕЅ(ввЫс) | NaOHШмвК | еєСѓ |
| D | ТШЛЏЧт(Cl2) | БЅКЭЪГбЮЫЎ | ЯДЦј |
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃКВЛЯъ ЬтаЭЃКЪЕбщЬт

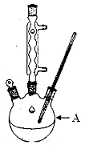

ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃКВЛЯъ ЬтаЭЃКЬюПеЬт
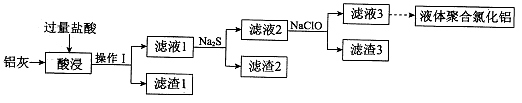


ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃКВЛЯъ ЬтаЭЃКЕЅбЁЬт
| AЃЎЫЎКЭЦћгЭЕФЛьКЯЮягУЗжвКЗНЗЈЗжРы |
| BЃЎЪЏЛвЫЎжааќИЁЕФЙЬЬхЬМЫсИЦгУЙ§ТЫЗНЗЈЗжРы |
| CЃЎЕтЫЎжаЕФЕтгУОЦОЋнЭШЁЗжРы |
| DЃЎввДМжаШмНтЮЂСПЪГбЮЃЌгУеєСѓЗНЗЈЗжРы |
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃКВЛЯъ ЬтаЭЃКЬюПеЬт
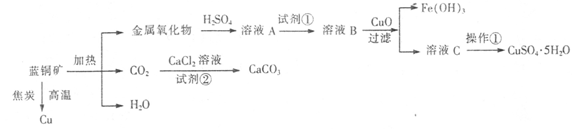
| Н№ЪєРызг | Fe3+ | Fe2+ | Cu2+ | |
| pH | ЧтбѕЛЏЮяПЊЪМГСЕэ | 1.9 | 7.0 | 4.7 |
| ЧтбѕЛЏЮяЭъШЋГСЕэ | 3.2 | 9.0 | 6.7 | |
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃКВЛЯъ ЬтаЭЃКЕЅбЁЬт
| AЃЎBaCl2ЁЂNaOHЁЂNa2CO3 |
| BЃЎBaCl2ЁЂNa2CO3ЁЂNaOH |
| CЃЎNaOHЁЂBaCl2ЁЂNa2CO3 |
| DЃЎNa2CO3ЁЂBaCl2ЁЂNaOH |
ВщПДД№АИКЭНтЮі>>
ЙњМЪбЇаЃгХбЁ - СЗЯАВсСаБэ - ЪдЬтСаБэ
КўББЪЁЛЅСЊЭјЮЅЗЈКЭВЛСМаХЯЂОйБЈЦНЬЈ | ЭјЩЯгаКІаХЯЂОйБЈзЈЧј | ЕчаХеЉЦОйБЈзЈЧј | ЩцРњЪЗащЮожївхгаКІаХЯЂОйБЈзЈЧј | ЩцЦѓЧжШЈОйБЈзЈЧј
ЮЅЗЈКЭВЛСМаХЯЂОйБЈЕчЛАЃК027-86699610 ОйБЈгЪЯфЃК58377363@163.com