
����Ŀ��ͬѧ�Ǵ�ɽ�ϲɼ���һ��ʯ��ʯ������ȡ80�˸���Ʒ���ж���ʵ�飨���������ڶ��չ����в������仯��,��÷�Ӧ������������m)�뷴Ӧʱ�䣨t)�Ĺ�ϵ���±���
��Ӧʱ��t�Ms | t0 | t1 | t2 | t3 | t4 | t5 | t6 |
��Ӧ����������m�Mg | 80 | 75 | 70 | 66 | 62 | 58 | 58 |
��ش��������⣺
(1)CaCO3��_____��Ԫ����ɣ����и�Ԫ�ص���������Ϊ_____
(2)��ʯ��ʯ��ȫ��Ӧ������CO2������Ϊ_____g
(3)���ʯ��ʯ��CaCO3������������д��������̡�______
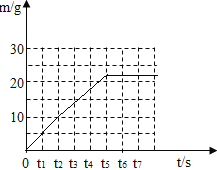
(4)������������ͼ�У���������ʱ���������������m)��ʱ��( t )�仯�����ߡ�____________
���𰸡��� 40% 22g 62.5% 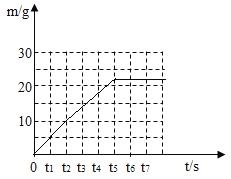
��������
��1��̼������ɸơ�̼��������Ԫ����ɵģ�̼��Ƶ���Է���������40+12+16��3=100��̼����и�Ԫ�ص���������Ϊ![]() ��100%=40%��
��100%=40%��
��2�����ڹ����������80g��Ϊ58g��������22g��˵�����ɵ����������Ϊ22g���ʴ�Ϊ��22g
��3���������ɵ����������Ϊ22g����˿��Ը������ɵ��������������������뷴Ӧ��̼��Ƶ��������ʴ�Ϊ��
�⣺����Ʒ�к�CaCO3������Ϊx��

![]()
x=50g
��Ʒ��CaCO3����������= ![]() ��100%=62.5%
��100%=62.5%
(4)�ɱ��е����ݿ�֪��t1��t2��t3��t4��t5ʱ���ɵĶ�����̼�������ֱ���5g��5g��4g��4g��4g��t5֮�����������塣
����ʱ���������������m����ʱ�䣨t���仯���������£�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������л�ʫ�ʡ�Ѱ�Ļ�����Ʒ����֮���������ӵġ��й�ʫ�ʴ�ᣩ����ȫ�����ʫ��֮��������ʫ��֮Ȥ������ʫ����ֻ�漰�����仯���ǣ�������
A. Ұ���ղ��������紵���������ù�ԭ���ͱ�
B. ������⣬ǧ����⣬����ѩƮ������![]() ѩ��
ѩ��
C. ������ǻ벻�£�Ҫ��������˼���ʯ������
D. ���ϵ���˿���������سɻ���ʼ��������![]() ���ʱ�ѱ����ѡ�
���ʱ�ѱ����ѡ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
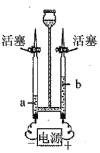
����Ŀ������ͼװ�ã��г�������ȥ���о���ȼ���ȼ��������ʵ����̣���ͨ��N2����W���Ҳಿ�ַ�����ˮ�У�a��b��������ȼ�գ���ͨ�������a�����ײ�ȼ�գ�b������ȼ�ա����ϣ�
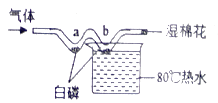
�����Ż��Ϊ40�棬P2O5����H2O��Ӧ��
��1������ȼ�յĻ�ѧ����ʽ��_____��
��2��ʵ������У���˵����ȼ��ȼ����Ҫ������������_____��
��3�����й��ڸ�ʵ���˵���У���ȷ����_____������ţ�
A ʪ���������հ���
B �ձ�����ˮ���������ṩ����
C ���У�a�����ײ�ȼ�յ�ԭ�����¶�û�дﵽ�Ż��
��4��С�ֲ����������ϵ�֪��þ���ڶ�����̼��ȼ������̼������þ��ͨ���Ը����ϵ��Ķ������ȼ�յ�����ʶΪ_____��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��д�����з�Ӧ�Ļ�ѧ����ʽ,��ע����Ӧ�Ļ�������
(1)̼��ȶ��ֽ�:____________________��___________��
(2)�и�����ʱ������ͭ��Һ����:_______________________��_____________��
(3)����������Ӧ�ⶨ�����������ĺ���:_____________________��______________.
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ѧ������ѧϰ��ѧ����Ҫ���ߣ����û�ѧ�������.
(1)���������������_____________
(2)����_____________
(3)����ˮ�Ļ�ѧ���ʵ���С������_____________
(4)���������������_____________
(5)����笠�����_____________
(6)�������þ��þԪ�صĻ��ϼ�_____________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��2018��������ˮ��������������Ϊ "Nature for water"��
(1)����ˮ��Դ��һ����Ҫ����ˮ����Ⱦ����һ����Ҫ______________��
(2)��ֱ��ˮ�����ɽ�����ˮ����Ϊ����ˮ�����ڲ�ʹ�õĻ���̿��Ҫ��____________���ã������г���_________��������ˮ��Ӳˮ������ˮ������___________�ķ�������ˮ��Ӳ�ȣ���������ɱ�������á�
(3)��ͼΪ���ˮ��ʵ��װ��ͼ��ͨ��һ��ʱ�������b���ռ�����������______����ʵ��˵��ˮ����___________ ��ɵġ�
(4)C1O2������ˮ���õ�����������ȡC1O2�Ļ�ѧ����ʽΪ��C12+2NaC1O2=2C1O2+2X��X�Ļ�ѧʽ__________��
(5)�����dz��л�ѧ�г�����ʵ�顣���˵���������______(�����)��
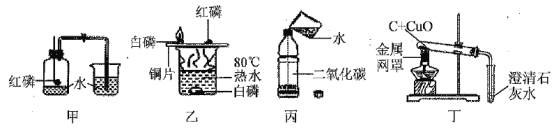
�ټ�ʵ���Ŀ���Dzⶨ�����������ĺ���
����ʵ�飬ˮ�еİ���û��ȼ�գ�����Ϊ�¶�û�дﵽ�Ż��
�۱�ʵ���ˮ����������ƿ�ǣ���������������������ƿ���ڰ���
�ܶ�ʵ�飬�ƾ����ϷŵĽ���������Ϊ������¶�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼ��ʾ���ĸ�ͼ��,����ȷ��ӳ��Ӧ�仯��ϵ����
A.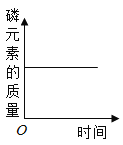 �����ܱ�������ȼ��,��Ԫ�ص������仯
�����ܱ�������ȼ��,��Ԫ�ص������仯
B.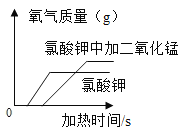 ��ͬ������������У�����һ�ݼ������������̣����������������仯
��ͬ������������У�����һ�ݼ������������̣����������������仯
C. ���ʷ�����ѧ�仯ǰ��,���ӵĸ����ı仯���
���ʷ�����ѧ�仯ǰ��,���ӵĸ����ı仯���
D.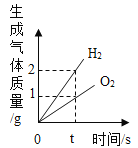 ˮ��ͨ�������,����������������������ϵ
ˮ��ͨ�������,����������������������ϵ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����Ҫ����д���пհס�
��1��������Ӧ����ѧ������գ�
��1��������_____
�ڸ�Ԫ��_____
��5�����������_____
�ܱ������������Ԫ�صĻ��ϼ�_____
���Ȼ�������_____��_____���������ɵ�
��2��ָ�����л�ѧ���������֡�2�������壺
��2H_____
��CO2_____
��Mg2+_____
��![]() ��_____
��_____
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��Ϊ�˲ⶨʵ�������������Ʒ�Ĵ��ȣ�ijѧϰС��ȡ2.5g����Ʒ��0.5g�������̻�ϣ����ȸû����t1ʱ��������ʲ��μӷ�Ӧ������ȴ������ʣ�������������ظ����ϲ��������γƵü���t1��t2�� t3��t4ʱ���ʣ��������������¼�������±�:
����ʱ�� | t1 | t2 | t3 | t4 |
ʣ���������/g | 2.48 | 2.34 | 2.04 | 2.04 |
����ϸ����ʵ�����ݣ��ش���������
��1����ȫ��Ӧ���������___________g
��2������Ʒ������ص���������_________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com