
| ||


 ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д�
ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д� ���������ν�ϵ�д�
���������ν�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
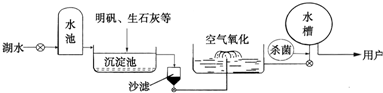
 ���µĿ������ྻ��ˮ�������Ӫ���������彡��ϢϢ��أ�����������ѧ��ѧ֪ʶ�ش��������⣺
���µĿ������ྻ��ˮ�������Ӫ���������彡��ϢϢ��أ�����������ѧ��ѧ֪ʶ�ش��������⣺
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ��2012?Ϋ�������µĿ������ྻ��ˮ�������Ӫ���������彡��ϢϢ��أ�����������ѧ��ѧ֪ʶ�ش��������⣺
��2012?Ϋ�������µĿ������ྻ��ˮ�������Ӫ���������彡��ϢϢ��أ�����������ѧ��ѧ֪ʶ�ش��������⣺
| ||
| ʳ�� | ��� | ���� | ������ | ţ�� |
| �����ʺ��� | 10% | 14% | 20% | 3% |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
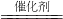 N2+4CO2���ڸ÷�Ӧ�л��ϼ۱仯��Ԫ��Ϊ______����дԪ�����ƣ���
N2+4CO2���ڸ÷�Ӧ�л��ϼ۱仯��Ԫ��Ϊ______����дԪ�����ƣ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012������ʡ����ݿ�Զ���п���ѧ��ģ�Ծ��������棩 ���ͣ������
| ���� | ϴ���� | ����� | ¯������ | ���ձ�ը�� | ����Һ |
| ��Ʒ ��ʽ |  |  |  |  |  |
| ��Ч�ɷֻ��� | ��ϴ���� | ���� | �������� | ��̼���� | ���� |

�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com