



 Ӧ�����������Ĵ���ѧ������ϵ�д�
Ӧ�����������Ĵ���ѧ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
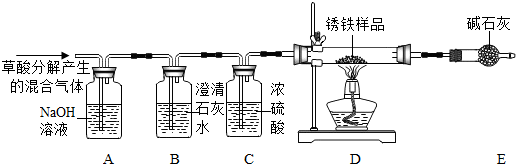
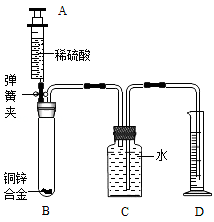
 ��2012?�人��ij��ѧС��������ͼ��ʾװ�òⶨͭп�Ͻ���Ʒ��п������������ͼ�й̶�װ������ȥ��
��2012?�人��ij��ѧС��������ͼ��ʾװ�òⶨͭп�Ͻ���Ʒ��п������������ͼ�й̶�װ������ȥ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| Ũ���� | ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ�п����� ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij��ѧС��������ͼ��ʾװ�òⶨͭп�Ͻ���Ʒ��п������������ͼ�й̶�װ��
����ȥ��
̽���������£�
1������ʵ��װ�ò����װ��������
����B�м�������Ϊm��ͭп�Ͻ��ĩ����ע����A��B
��������ϡ���ᣬ��B�в����������������ʱ���н����ɼУ�
�����Ƴ�D�е��ܣ�ȷ��ȡ����¼��Ͳ��ˮ�������
�����˵Ȳ�����ȷ��������¼B��ʣ��������ʵ�
������
IV����С��ļ�ͬѧ���ݷ�Ӧǰ��B�й������ʵ�������
����Ʒ��п��������������ͬѧ�϶���Ͳ�ڲ��ˮ�����
��Ϊ��Ӧ�������������������ø������ڳ����µ��ܶȣ����ݻ�ѧ����ʽ������ؼ��㣬�õ���Ʒ��п������������
��ش��������⣺
��1�� B�з�����Ӧ�Ļ�ѧ����ʽ��
��
��2�����У���ȷ�IJ���˳���ǣ����ˡ� �� ��������
��3��ͨ������������ѧС�鷢����ͬѧ�������ݲ��ɿ�����ɸ����ݲ��ɿ���ԭ����Լ�������Ӱ���ǣ�
 ��II�У��Ƴ�D�еĵ�����������ˮ�����¼�����ƫС��
��II�У��Ƴ�D�еĵ�����������ˮ�����¼�����ƫС��
�� �����¼�����ƫ ��
����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
ij��ѧС��������ͼ��ʾװ�òⶨͭп�Ͻ���Ʒ��п������������ͼ�й̶�װ������ȥ��̽���������£�
1������ʵ��װ�ò����װ��������
����B�м�������Ϊm��ͭп�Ͻ��ĩ����ע����A��B��������ϡ���ᣬ��B�в����������������ʱ���н����ɼУ������Ƴ�D�е��ܣ�ȷ��ȡ����¼��Ͳ��ˮ�������
�����˵Ȳ�����ȷ��������¼B��ʣ��������ʵ�������
IV����С��ļ�ͬѧ���ݷ�Ӧǰ��B�й������ʵ�����������Ʒ��п��������������ͬѧ�϶���Ͳ�ڲ��ˮ�����
��Ϊ��Ӧ�������������������ø������ڳ����µ��ܶȣ����ݻ�ѧ����ʽ������ؼ��㣬�õ���Ʒ��п��������������ش��������⣺
��1�����У���ȷ�IJ���˳���ǣ����ˡ� �� ��������
��2��ͨ������������ѧС�鷢����ͬѧ�������ݲ��ɿ�����ɸ����ݲ��ɿ���ԭ����Լ�������Ӱ���ǣ�
��II�У��Ƴ�D�еĵ�����������ˮ�����¼�����ƫС��
�� ��
����
��3���ճ�������Ϊ����ʹ��ͨ���������ƳɺϽ�,��DZ�Ϊ��о��ͭ��һԪ��Ϊ��о������Ni���Ͻ���������Ӳ���õ��IJ��϶��� ����
‚ѡ������Ӳ�ҵIJ��ϲ���Ҫ���ǵ������� ������ţ���
A. �����ĵ����� B. ��������ʴ��
C. ������Ӳ�� D. �����۸���Ӳ����ֵ���Ǻ϶�
ƒ����Ӳ���漰��Fe��Cu��Al��Ni����������֪Ni�ܹ��������·�Ӧ��
Ni+ H2SO4= Ni SO4+ H2�� Fe+ Ni Cl 2= Ni + Fe Cl 2
���ֽ��������ǿ������˳��Ϊ ����Ԫ�ط��ű�ʾ����

�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com