
����Ŀ��̼Ԫ��������������ʵĻ���Ԫ�أ��ش����к�̼Ԫ�����ʵ��й����⣮
(1)��������̼�������Ҫ��Ϊ�˼���_______���ŷ�����
(2)����CO2���ó���ʯ��ˮ��д���÷�Ӧ�Ļ�ѧ����ʽ_________________��
(3)��Ȼ��(��Ҫ�ɷ���CH4)��ȼ��____________________(�û�ѧ����ʽ��ʾ)��
(4)Ϊ������������CO2���ŷţ���ѧ�ҽ�CO2��H2�ڴ����ͼ��ȵ������·�Ӧ��ת��Ϊˮ�ͼ��飬�÷�Ӧ�Ļ�ѧ����ʽΪ_________________________________������˵����������CO2�ĺ���Խ��Խ���������Ƿ�ͬ������˵��(����������������)_____��������________________________��
(5)���ʯ��ʯī���������ʲ���ϴ���Ҫԭ����____________________��
(6)�ڹ�ҵ�Ͽ�����CO��NaOHͨ�����Ϸ�Ӧ�Ʊ�HCOONa(������)���÷�Ӧ�Ļ�ѧ����ʽΪ________________________________��
���𰸡� CO2 CO2+Ca(OH)2�TCaCO3��+H2O CH4+2O2![]() CO2+2H2O CO2+4H2
CO2+2H2O CO2+4H2![]() CH4+2H2O �� ��������ж�����̼�ĺ���̫�ͣ���Ӱ��ֲ��Ĺ������ ̼ԭ�ӵ����з�ʽ��ͬ CO+NaOH
CH4+2H2O �� ��������ж�����̼�ĺ���̫�ͣ���Ӱ��ֲ��Ĺ������ ̼ԭ�ӵ����з�ʽ��ͬ CO+NaOH![]() HCOONa
HCOONa
����������1������̼�����������������������ģ���Ҫ��Ϊ�˼��ٶ�����̼���ŷ���
��2��������̼���������Ʒ�Ӧ����̼��Ƴ�����ˮ����Ӧ�Ļ�ѧ����ʽΪ��CO2+Ca(OH)2�T CaCO3��+H2O��
��3�������������ڵ�ȼ�������·�Ӧ���ɶ�����̼��ˮ����ѧ����ʽ��ʾΪ��CH4+2O2![]() CO2+2H2O��
CO2+2H2O��
��4��������̼�������ڴ����ͼ��ȵ������·�Ӧ����ˮ�ͼ��飬��ѧ����ʽΪ��CO2+4H2![]() CH4+2H2O������˵����������CO2�ĺ���Խ��Խ��������ͬ������˵���������ǣ���������ж�����̼�ĺ���̫�ͣ���Ӱ��ֲ��Ĺ�����ã�
CH4+2H2O������˵����������CO2�ĺ���Խ��Խ��������ͬ������˵���������ǣ���������ж�����̼�ĺ���̫�ͣ���Ӱ��ֲ��Ĺ�����ã�
��5�����ʯ��ʯī���������ʲ���ϴ���Ҫԭ����̼ԭ�ӵ����з�ʽ��ͬ��
��6���ڹ�ҵ�Ͽ�����CO��NaOHͨ�����Ϸ�Ӧ�Ʊ�HCOONa�������ƣ����÷�Ӧ�Ļ�ѧ����ʽΪ��CO+NaOH![]() HCOONa��
HCOONa��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����л�ѧ����ʽ����ȷ��ʾ�������ݵ���
A����Ƭ�뵽ϡ���2Fe+3H2SO4=Fe2(SO4)3 + 3H2��
B���������Ʊ�����2NaOH + CO2 = Na2CO3 + H2O
C��ʵ�����Ʊ�CO2��CaCO3 ���� CaO+CO2��
D����˿��������ȼ����4Fe + 3O2 ��ȼ 2Fe2O3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���������о������ö����������������Ҫ���壮
(1)��������Ǧ�Ͻ𣬰�Ǧ���������ƳɺϽ����ҪĿ����_________��
(2)����Ʒ�������γ�������������Ĥ���Ӷ���ֹ����һ�����������������������������ᣮд����������ϡ���ᷴӦ�Ļ�ѧ����ʽ____________________��
(3)��װ�������ļ���ƿ�����ڼ������ۻ��Ľ������ϣ��ɹ۲쵽��������_________________���÷�Ӧ�Ļ�ѧ����ʽ��_____________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��(1)������ѧ��ѧ֪ʶ�����������⣺
���������γ���������Ĥ�Ļ�ѧ����ʽ__________�� ��![]() ___��
___��
(2) ��������(NaN3)���㷺Ӧ����������ȫ���ҡ��������Ƶ��Ʊ�������������Һ̬����Ӧ��NaNH2���ٽ�NaNH2��N2O��Ӧ������NaN3���÷�Ӧ�Ļ�ѧ����ʽΪ2NaNH2 + N2O=NaN3 + NaOH + NH3����ش��������⣺
��N2O��������____________��Һ̬������________(�����������������������)��
��NaNH2�е�Ԫ�صĻ��ϼ�Ϊ____________������H��N��NaԪ�ص�������Ϊ_____��
��NaN3�е�������Ϊ______��������ײ����30������NaN3Ѹ�ٷֽ�ΪNa��N2���䷴Ӧ�Ļ�ѧ����ʽΪ________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ij��ȤС��Ϊ����������������Һ������������Һ�����������ͼ��ʾ������ʵ�鷽�����밴Ҫ��ش����⣮
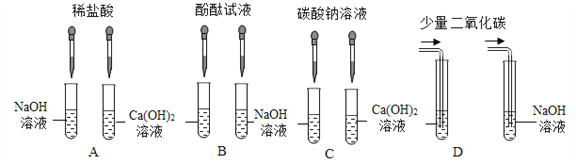
(1)����ʵ�鷽�����ܴﵽʵ��Ŀ����_______(����ĸ���)��
(2)C��ʵ���з�����Ӧ�Ļ�ѧ����ʽΪ___________________________��
������̽����ʵ�������С���ͬѧ��A��B��C��D����ʵ���֧�Թ��е����ʵ���ͬһ���ձ��У����յõ���ɫ������Һ��
���������ۡ�
���ձ����ܸ�ϡ���ᷢ����ѧ��Ӧ��������_______��(��̪����)��
�����յõ�����Һ�У�һ�����е�������_____________(�ѧʽ����ͬ)��һ�������е�������________�����ܺ��е�������_______��
��ʵ����֤��Ϊ����֤���յõ�����Һ�п��ܺ��е������Ƿ���ڣ�С���ͬѧȡ�����ձ��е���Һ���Թ��У���������μ���̼������Һֱ���������۲쵽________��֤�����и����ʣ�
����չ���졿С���ͬѧ��D��ʵ�鷽���еĶ�����̼���ɶ������ﵽͬ��ʵ��Ч��������������������Ʒ�Ӧ�Ļ�ѧ����ʽΪ______________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʯ��ʯ�������������Ȼ��Ƶȶ��ֻ�����Ʒ��ԭ�ϡ�ij�о���ѧϰС��Ϊ�˲ⶨ���ؿ�ɽʯ��ʯ��̼��Ƶ�����������ȡ���˿�ʯ��Ʒ����ȡϡ����200g��ƽ���ֳ�4�ݣ�����ʵ�飬������£�
ʵ �� | 1 | 2 | 3 | 4 |
������Ʒ����/g | 5 | 10 | 15 | 20 |
����CO2������/g | 1.76 | 3.52 | 4.4 | m |
(1)�ڼ��η�Ӧ�п�ʯ��ʣ�ࣿ_________��
(2)����mֵ��________��
(3)�Լ�������ʯ��ʯ��̼��Ƶ���������Ϊ_________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���ڸ�ðҩ�м�����������PPA����ҩ��������Ը��Ƹ�ð֢״������ҩ���������Ѫ�ܼ�������������Ӱ�졣2000�����������Ҫ���ֹʹ�����ͳ��ۺ�����PPA���ĸ�ðҩ����PPA���Ļ�ѧʽΪC9H13NO���ɴ˿�֪��
(1)PPA����_______��Ԫ����ɣ�һ��PPA�����й�����______��ԭ�ӡ�
(2)PPA��̼Ԫ������Ԫ�ص�������Ϊ________________________
(3)PPA�е�Ԫ�ص���������Ϊ__________��
(4)15.1�˵�PPA���е�Ԫ�ص�����Ϊ___________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��Ϊ��ȷ��CH4��H2��CO (���¼��������)���ֿ�ȼ�����壬�ֱ�������������ȼ�գ���ȼ�պ����ɵ���������ͨ����ͼ��ʾ��װ�ã��ش��������⣮����ܰ��ʾ��Ũ��������ˮ�ԣ���ˮ���������ӣ���
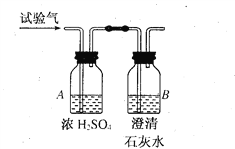
��1����װ��A���������ӣ�B���������䣬����������________��ȼ�յĻ�ѧ����ʽΪ_________________________________��
��2����װ��A���������䣬B���������ӣ�����������_________����Bƿ�еķ�Ӧ����ʽ��________________________________��
��3����װ��A��B�����������ӣ�����������_________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��2010�괺�ҹ����ϴֵ����������صĺ��֣������ڿ������ֵ�ͬʱ����Ӧ��������ˮ��������ϧˮ��Դ����ش��������⣺
(1)����������ˮ���Ǵ���ˮ������Ϊ����ˮ�Ļ�ѧ���ʵ���С����___________������ѡ������ȷ��ˮ������Ԫ�غ���Ԫ����ɵ�ʵ����_________(�����)��
A��ˮ������ B��ˮ������ C��ˮ�ĵ�� D��ˮ�ľ���
(2)��ͼ���ҹ����ֵ����˾�ˮ��ͼ�����п��Կ����ӱ��Ǽ���ȱˮ��ʡ�������������Ͻ�Լ��ˮ����____________(�����)��
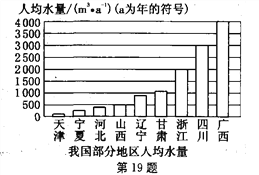
A��ʹ�ý�ˮ��ͷ
B��������ˮϴ��
C������ࡢ�ι����ũ���スˮ
D��ʹ�ò���ϵر�עˮ�߳��ܡ���ˮ����ˮ��Ϊ165L/�εļ���ϴ�»�
(3)ʵ��������һ������MgCl2��CaCl2��Ӳˮ���±���ij��ȤС���������Ӳˮ��ʵ�鷽��������һ��������±���
��ѡ�õ�ҩƷ�У�Ca(OH)2��Һ��NaOH��Һ��Na2CO3��Һ������ˮ��
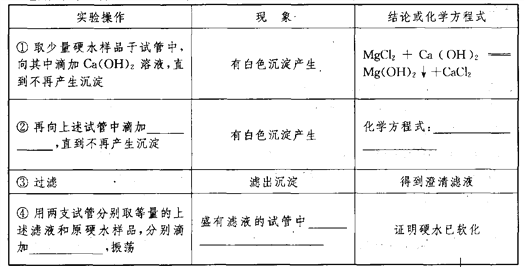
____________ ____________ ____________ ____________
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com