
��1��(8��) ij��ɫ����Һ���ܴ���Na����Fe3����Ba2����NO3����CO32����HCO3����SO42���еļ������ӣ��������²�����
��ȡ��������Һ����CaCl2��Һ�����������μ����������ɫ��ζ�����塣
����ȡ����Һ�μ�һ������NaOH��Һ�а�ɫ�������ɡ�
�Իش��������⣺
�� ����Һ��һ�����ڵ�������__________��һ�������ڵ�����__________��
�� ��һ�����ӷ���ʽ��ʾ�����ڵ�ʵ������
______________________________________________________��
��2����4�֣����и����е�������������Һ�еķ�Ӧ������ͬһ���ӷ���ʽ��ʾ���ǣ� ������д�������ӷ�Ӧ����ʽ��___________________________ __________________��
__________________��
| A��������ռ���Һ�����������������Һ |
B���� ����Һ�����ᡢʯ��ʯ������ ����Һ�����ᡢʯ��ʯ������ |
| C���Ȼ�����Һ����������Һ�����ᱵ��Һ������ |
| D��̼�����Һ������������Һ��̼�����Һ������������Һ |
 ���100��1�ž�ϵ�д�
���100��1�ž�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2012�������ѧ�ڻ�ѧһ�ָ�ϰ����ѧ�����е��������硷ר���ۺϲ��ԣ��ս̰棩 ���ͣ�ʵ����
(8��)��ͼ�������Լ�ƿ��ǩ�ϵ����ݣ�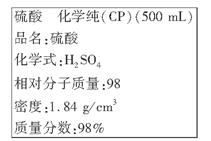
(1)����������ʵ���Ũ����________mol/L��
(2)ij��ѧ��ȤС������������ʵ�ʵ��̽��ʱ����Ҫ240 mL 4.6 mol/L��ϡ���ᣬ����Ҫȡ________mL�ĸ����ᡣ�������������ϡ�͵�ʵ�������_____________________��
(3)������4.6 mol/L��ϡ����Ĺ����У����������������Һ���ʵ���Ũ���к�Ӱ��(�ƫ�ߡ�����ƫ�͡�����Ӱ�족)
��δ����ȴ���Ƚ���Һע������ƿ��________��
������ƿ��1 mol/Lϡ������ϴ________��
�۶���ʱ���ӹ۲�Һ��________��
(4)�����£�ijѧ��ȡ�������Լ�ƿ�е�������һ�ྻ�Թ��У��Ӽ�Ƭ���������Ƭ����û�����ݲ�����Ҳδ������Ƭ�ܽ⣬������ͬѧ����ԭ��______________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�����ʡ����ػ�����ѧ��һ12���¿���ѧ�Ծ� ���ͣ������
10�֣�ij��ѧ��ȤС���ijƷ��������Ħ�����ɷּ��京����������̽����
������ϣ�������Ħ������̼��ơ�����������ɣ������������ɷ���������ʱ���������ɡ�
��.Ħ���������������Ķ��Լ���
ȡ����������Ʒ����ˮ�ɷֽ��衢���ˡ�
��1���������м������NaOH��Һ�����ˡ�����������NaOH��Һ��Ӧ�����ӷ���ʽ��__________________________ ___ ______��
��2������1��������Һ����ͨ�����������̼���ټ������ϡ���ᡣ�۲쵽��������______________________ ___ ______��
��.������Ʒ��̼��ƵĶ����ⶨ
������ͼ��ʾװ�ã�ͼ�мг�������ȥ������ʵ�飬��ַ�Ӧ�ⶨC�����ɵ�BaCO3������������ȷ��̼��Ƶ�����������
����ʵ����̻ش��������⣺
��3��ʵ����������������ͨ������������ó��˿ɽ���B��C�еķ�Ӧ���⣬���У�____________________ _____________��
��4��C�з�Ӧ����BaCO3�Ļ�ѧ����ʽ��_______________________ _________��
��5��ʵ����ȷ��ȡ8.00g��Ʒ���ݣ��������βⶨ�����BaCO3ƽ������Ϊ3.94g������Ʒ��̼��Ƶ���������Ϊ_________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013ѧ�����ʡ��ˮ�и�����һ���¿���ѧ�Ծ��������棩 ���ͣ�ʵ����
(8��)��ͼ�������Լ�ƿ��ǩ�ϵ����ݣ�

(1)����������ʵ���Ũ����________mol/L��
(2)ij��ѧ��ȤС������������ʵ�ʵ��̽��ʱ����Ҫ240 mL 4.6 mol/L��ϡ���ᣬ����Ҫȡ________mL�ĸ����ᡣ�������������ϡ�͵�ʵ�������_____________________��
(3)������4.6 mol/L��ϡ����Ĺ����У����������������Һ���ʵ���Ũ���к�Ӱ��(�ƫ�ߡ�����ƫ�͡�����Ӱ�족)
��δ����ȴ���Ƚ���Һע������ƿ��________��
������ƿ��1 mol/Lϡ������ϴ________��
�۶���ʱ���ӹ۲�Һ��________��
(4)�����£�ijѧ��ȡ�������Լ�ƿ�е�������һ�ྻ�Թ��У��Ӽ�Ƭ���������Ƭ����û�����ݲ�����Ҳδ������Ƭ�ܽ⣬������ͬѧ����ԭ��______________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ��߿���ѧһ�ָ�ϰ����Һ�е����ӷ�Ӧ��ר���ۺϲ��ԣ��ս̰棩 ���ͣ�ʵ����
(8��)���к͵ζ����ⶨij�ռ���Ʒ�Ĵ��ȣ��Ը���ʵ��ش��������⣺
(1)ȷ����8.2 g�������������������ʵ���Ʒ�����500 mL������Һ������ʱ����Ʒ�ɷ���________(������ĸ)������
A��С�ձ��С�������B���ྻֽƬ�ϡ�������C��������
(2)�ζ�ʱ����0.2000 mol��L��1���������ζ�������Һ������ѡ��________(������ĸ)��ָʾ����
A�����ȡ��� B��ʯ���
C����̪���� D������
(3)�ζ������У��۾�Ӧע��______________��������̨�ϵ�һ�Ű�ֽ����Ŀ����________________________________________________________________________
________________________________________________________________________��
(4)�����±����ݣ����㱻���ռ���Һ�����ʵ���Ũ����________mol��L��1���ռ���Ʒ�Ĵ�����________��
|
����� |
������Һ ���(mL) |
������� |
|
|
�ζ�ǰ�Ŀ̶� (mL) |
�ζ���Ŀ̶� (mL) |
||
|
��һ�� |
10.00 |
0.40 |
20.50 |
|
�ڶ��� |
10.00 |
4.10 |
24.00 |
(5)����ʵ�������Եζ��������ʲôӰ��(�ƫ�ߡ�����ƫ�͡�����Ӱ�족)?
�ٹ۲���ʽ�ζ���Һ��ʱ����ʼ���ӣ��ζ��յ�ƽ�ӣ���ζ����________��
��������ƿ�ô���Һ��ϴ��Ȼ���ټ���10.00 mL����Һ����ζ����________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com