
��15�֣� A��B��C��X����ѧ��ѧ�������ʣ����ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��������������ֲ�ͬ����ش�
��1����A��B��C�о���ͬһ�ֳ�������Ԫ�أ���Ԫ���� C������������ʽ���ڣ���A��C��ˮ��Һ��Ͽɵ�B�İ�ɫ��״������
�� A�к��еĽ���Ԫ�ص�ԭ�ӽṹʾ��ͼΪ ��
�� �ý���Ԫ�صĵ�����ij��ɫ�������ڸ����·�Ӧ�������ں������켰�����ƣ���֪��1mol�õ�����ȫ��Ӧ�����¶Ȼָ���298Kʱ��������QkJ����д���÷�Ӧ���Ȼ�ѧ��Ӧ����ʽΪ ��
��2����AΪ�л��75%��A��Һ����Ϊ�����������³�ѹ��B��C��Ϊ��ɫ���壬C��һ�ֳ�������������A�Ľṹ��ʽΪ�� ������ԭ���ԭ������B��X�ֱ�ͨ����A�Ƴɵ�����缫����20%��30%��KOH��Һ��Ϊ�������Һ��������ɻ�ѧ��Դ���õ�طŵ�ʱ�������缫��ӦʽΪ ��
��3����A��B��C����ɫ��Ӧ���ʻ�ɫ��ˮ��Һ��Ϊ���ԡ�
���û�ѧ����ʽ����C��Һ�ʼ��Ե�ԭ�� ��
�ڽ�4.48 L����״���£�Xͨ��100 mL3 mol/L A��ˮ��Һ����Һ������Ũ���ɴ�С��˳��Ϊ ��
����Ȼ���д���B��C��H2O��һ�������ᾧ���ɵĹ��塣ȡһ�����ù�������ˮ���100 mL��Һ�������Һ�н��������ӵ�Ũ��Ϊ0.5 mol/L����ȡ��ͬ�����Ĺ�����������أ�ʣ����������Ϊ___________________��
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 A��B��C��X����ѧ��ѧ1�г�����4�����ʣ����Ǿ��ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��ʾ������������������1�ش��������⣺
A��B��C��X����ѧ��ѧ1�г�����4�����ʣ����Ǿ��ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��ʾ������������������1�ش��������⣺
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
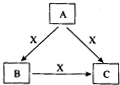 A��B��C��X����ѧ��ѧ�������ʣ�ת����ϵ��ͼ��ʾ����������²�ͬ����ش����⣺
A��B��C��X����ѧ��ѧ�������ʣ�ת����ϵ��ͼ��ʾ����������²�ͬ����ش����⣺
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 A��B��C��X����ѧ��ѧ�������ʣ����ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��ʾ����������²�ͬ����ش�
A��B��C��X����ѧ��ѧ�������ʣ����ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��ʾ����������²�ͬ����ش��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
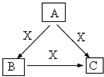 A��B��C��X����ѧ��ѧ�������ʣ����ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��������������ֲ�ͬ����ش�
A��B��C��X����ѧ��ѧ�������ʣ����ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��������������ֲ�ͬ����ش�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 A��B��C��X����ѧ��ѧ�г��������ʣ�����֮���ת����ϵ��ͼ��ʾ�����ֲ�������ȥ������ش��������⣺
A��B��C��X����ѧ��ѧ�г��������ʣ�����֮���ת����ϵ��ͼ��ʾ�����ֲ�������ȥ������ش��������⣺�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com