
| ʵ����� | KClO3���� | ������������ | �������� |
| �� | 1.2g | ���������� | |
| �� | 1.2g | CuO 0.5g | |
| �� | 1.2g | MnO2 0.5g |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
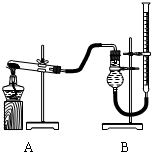
 ��2011?�人ģ�⣩ijʵ��С������ȡ����ͭ��֤������ͭ�ܼӿ�����صķֽ⣬����������ʵ�飺
��2011?�人ģ�⣩ijʵ��С������ȡ����ͭ��֤������ͭ�ܼӿ�����صķֽ⣬����������ʵ�飺| ʵ����� | KClO3���� | ������������ | �������� |
| �� | 1.2g | ���������� | |
| �� | 1.2g | CuO 0.5g | |
| �� | 1.2g | MnO2 0.5g |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ʵ����� | KClO3���� | ������������ | �������� |
| �� | 1.2g | ���������� | |
| �� | 1.2g | CuO 0.5g | |
| �� | 1.2g | MnO2 0.5g |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(һ)��ȡ����ͭ
�ٳ�ȡ
(1)����ʵ�鲽������Ҫʹ�ò���������_______________(��ʵ�����)������٣�������ĥ��������������������_______________��
(2)�������ϴ�ӳ����IJ�����_________________________________________��
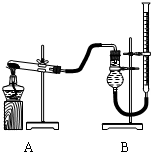
(��)֤������ͭ�ܼӿ�����صķֽⲢ��������̵Ĵ�Ч�����бȽ�����ͼװ�ý���ʵ�飬ʵ��ʱ��������25 mL����Ϊ����������Ӱ��ʵ������ؾ��Ѻ��ԣ�������ݼ��±�:
ʵ����� | KClO3���� | ������������ | �������� |
�� | ���������� |
| |
�� | CuO |
| |
�� | MnO2 0.5 |
|
�ش���������:
(3)����ʵ���еġ��������ݡ�ָ_______________��
(4)��Ҫ֤��ʵ����и�������ռ���������O2���ɴ������ռ��������õ��ɼм�סB���齺�ܣ���ȥ������ϵ�����Ƥ����_______________��
(5)Ϊ̽��CuO��ʵ������Ƿ�������ã��貹������ʵ��(����д���������):
a._______________��b.CuO�Ļ�ѧ������û�иı䡣
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�콭��ʡ�����ѧ�����β��Ի�ѧ�Ծ����������� ���ͣ�ʵ����
��14�֣�ijʵ��С������ȡ����ͭ��֤������ͭ�ܼӿ�����صķֽ⣬����������ʵ�飺
��һ����ȡ����ͭ
�ٳ�ȡ2 gCuSO4��5H2O��ϸ�����ձ�����10 mL����ˮ�ܽ⣻
��������CuSO4��Һ����μ���NaOH��Һ��ֱ�����ٲ���������Ȼ�����û����ת�Ƶ���������������ȫ����Ϊ��ɫ��
�۽���������û������ˡ�ϴ�ӣ����ɺ���ϸ���á�
�ش��������⣺
������ʵ�鲽������Ҫʹ�ò���������_______________����ʵ����ţ�������١�������ĥ��������������������___________________��
�Ʋ������ϴ�ӳ����IJ�����______________________________________________
__________________________________________________________��
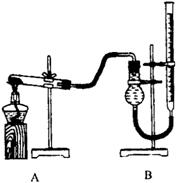
������֤������ͭ�ܼӿ�����صķֽⲢ��������̵Ĵ�Ч�����бȽ�
����ͼװ�ý���ʵ�飬ʵ��ʱ��������25 mL����Ϊ����������Ӱ��ʵ������ؾ��Ѻ��ԣ�������ݼ��±���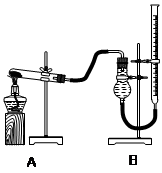
| ʵ����� | KClO3���� | ������������ | �������� |
| �� | 1.2 g | ���������� | |
| �� | 1.2 g | CuO 0.5 g | |
| �� | 1.2 g | MnO2 0.5 g | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�꽭��ʡ��������ѧ��һ��ѧ�����п��Ի�ѧ�Ծ����������� ���ͣ�ʵ����
ijʵ��С������ȡ����ͭ��֤������ͭ�ܼӿ�����صķֽ⣬����������ʵ�飺
��һ����ȡ����ͭ
�ٳ�ȡ2 gCuSO4��5H2O��ϸ�����ձ�����10 mL����ˮ�ܽ⣻
��������CuSO4��Һ����μ���NaOH��Һ��ֱ�����ٲ���������Ȼ�����û����ת�Ƶ���������������ȫ����Ϊ��ɫ��
�۽���������û������ˡ�ϴ�ӣ����ɺ���ϸ���á�
�ش��������⣺
������ʵ�鲽������Ҫʹ�ò���������_______________����ʵ����ţ�
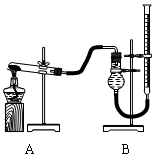
������֤������ͭ�ܼӿ�����صķֽⲢ��������̵Ĵ�Ч�����бȽ�������ͼװ�ý���ʵ�飬ʵ��ʱ��������25 mL����Ϊ����������Ӱ��ʵ������ؾ��Ѻ��ԣ�����������£�
| ʵ����� | KClO3���� | ������������ | �������� |
| �� | 1.2 g | ���������� | |
| �� | 1.2 g | CuO 0.5 g | |
| �� | 1.2 g | MnO2 0.5 g | |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com